Abstract
Applying wide-angle X-ray scattering method, thermomechanical analysis, and differential scanning calorimetry, the structural organization and properties of nanocomposites formed by chemical reduction of Сu2+ cations in the interpolyelectrolyte–metal complex (pectin–Cu2+–polyethyleneimine) under the influence of a constant magnetic and electric fields have been studied. It has been found that the chemical reduction of Cu2+ cations in the interpolyelectrolyte–metal complex bulk under constant electric and magnetic fields leads to formation of nanocomposite consisting of interpolyelectrolyte complex, including pectin–polyethyleneimine and nanoparticles of the metal Cu phase, whereas nanocomposite with Cu/Cu2O nanoparticles is formed in original state (without any field). It was observed that, under constant field, nanocomposites obtained have higher structural glass-transition temperatures and thermal stability.
Similar content being viewed by others
Background
In the last decade, considerable attention is paid to the scientific researches dealing with polymer nanocomposites, filled with nanoparticles of different metals or metal oxides [1–3].
Metallo-containing compounds can provide polymer materials with special optical, electrical, magnetic, and mechanical properties as well as catalytic activity [4–8]. The capability of functional groups on polyelectrolytes to bind metal ions offers a possibility for their application as sorbing agents, ion-exchange materials, components of selective membranes [8–10], or as precursors for preparation of polymer–inorganic hybrids via reduction or precipitation of metal ions [8–12]. Polymer–inorganic nanocomposites are important candidates for construction of photonic devices, band-pass filters, components of nonlinear optical systems, optical limiters, elements of microcircuit chips, etc. [5, 6, 13]. Polyelectrolyte-based materials, including ultrafine particles of silver and noble metals, exhibit antibacterial properties and are therefore promising objects for application in medicine [13–17].
The current methods of preparing polymer–metal nanocomposites are mainly multistage, for example, the synthesis of metal or metal oxide nanoparticles followed by their introduction into the polymer bulk [2, 3, 18]. The above approach has a disadvantage due to difficulties in providing a uniform nanoparticle distribution throughout the polymer matrix. An alternative method of nanocomposite synthesis is the reduction of metal ions (Мen+) in interpolyelectrolyte–metal complexes formed via the introduction of metal salts into an interpolyelectrolyte complex based on two oppositely charged polyelectrolytes [8, 19, 20]. This method allows the preparation of nanocomposites with uniform distributions and controlled nanoparticle sizes in the polymer matrix.
The structuring and thermomechanical properties of pectin–Cu2+–polyethyleneimine interpolyelectrolyte–metal complexes (IMC) and related nanocomposites were studied previously [21]. It was found that the chemical reduction of Cu2+ cations in the IMC bulk with the use of NaBH4 results in interpolyelectrolyte complexes (IPEC) and Cu/Cu2O nanocomposites, and, at the molar ratio BH4 −:Cu2+ = 6, the structure of the Cu metal phase manifests itself completely. As it was earlier shown [22], reduction of Cu2+ cations by NaBH4 in the triple polyelectrolyte–metal complexes, influencing by a constant magnetic field allows physico-mechanical properties of nanocomposites to be enhanced.
So, the aim of this work is to investigate the effect of constant electric and magnetic fields on the structural organization and thermomechanical and thermophysical properties of nanocomposites prepared involving a natural and synthetic polymers—pectin, polyethyleneimine, and Cu nanoparticles, formed from pectin–Cu2+–polyethyleneimine interpolyelectrolyte–metal complexes.
Methods
To obtain the IPEC, the IMC, pectin–Cu2+–polyethyleneimine, and nanocomposites of IPEC–Cu/Cu2O or IPEC–Cu, the following reagents were used: anionic polyelectrolyte citrus pectin (Cargill Deutschland GmbH, Germany) with М = 3 × 104, cationic polyelectrolyte anhydrous branched polyethyleneimine (PEI) (Aldrich) with М n = 1 × 104 and М w = 2.5 × 104, copper(II) sulfate pentahydrate (CuSO4 × 5H2O) (Aldrich) with М = 249.69, and sodium borohydride (NaBH4) (Aldrich) with М = 37.83.
IPEC samples were formed via mixing of 5 % aqueous solutions of pectin and PEI taken at a molar ratio of 1:1 at Т = 20 ± 2 °С. IPEC as films were prepared via pouring onto PTFE plates and drying up to constant masses at the same temperature. Dry IPEC films were washed in distilled water up to neutrality and dried repeatedly at 20 °С up to constant masses. The resulting films were 100-μm thick.
IMC samples were prepared via immersion of IPEC films into an aqueous solution of CuSO4 with a concentration of 0.1 mol/L at Т = 20 ± 2 °С for 24 h. The colorless IPEC films became dark blue.
The adsorption capacities of films, А (mmol/g), were calculated through the formula [23]
where m is the mass of the adsorbent, V is the solution volume, and c in and c eq are the initial and the equilibrium concentrations of copper ions. For IMC films, А = 2.9 mmol/g.
The chemical reduction of Cu2+ cations in the IMC was conducted with NaBH4 (a molar ratio of BH4 −:Cu2+ = 6.0) in an alkaline medium (pH 10.8) in a solvent mixture of water–isopropanol (4:1 vol.%) at Т = 20 ± 2 °С for 3 h (until the release of gaseous bubbles ceased). The concentration of NaBH4 in the aqueous alcohol solution was 0.1 mol/L. As a result of the reduction, IMC films changed color from blue to dark brown that confirms the formation of Cu2О nanoparticles in the polymer matrix [19].
The reduction of Cu2+ cations in the IMC was performed both in the absence and in the presence of a constant electric field (E = 1 × 106 V/m) for 3 h between the plates of a plane capacitor and as well in constant magnetic field (B = 0.2 T) for 3 h between the poles of electromagnet. In both cases, film surface was placed perpendicularly to the field force lines (Т = 20 ± 2 °С). Values of the electric field intensity, magnetic field induction, and reduction time for Cu2+ cations were selected as optimal in our experiment.
The features of the amorphous and amorphous–crystalline structuring of the IPEC (pectin–PEI), the IMC (pectin–Cu2+–PEI), and nanocomposites of IPEC–Cu/Cu2O or IPEC–Cu were studied by wide-angle X-ray diffraction on a DRON-4-07 diffractometer (scientific-production company “Burevestnik,” Russia), whose X-ray optical scheme was used to “pass” primary-beam radiation through samples. X-ray diffraction studies were performed at Т = 20 ± 2 °С in CuК α radiation monochromated with a Ni-filter.
The size of the Cu/Cu2O nanoparticles and their distribution in the polymer matrix were examined with a JEM-1230 transmission electron microscope (JEOL, Japan) at a resolution of 0.2 nm.
Thermomechanical studies of polymer systems were conducted using the penetration method in the mode of a uniaxial constant load (σ = 0.5 MPa) on a UIP-70M device (central design engineering bureau of the special instrument making of the National Academy of Sciences of Russia). Linear heating of samples was performed at a rate of 2.5 °С/min in the temperature range from −100 to +350 °С.
Thermophysical researches were performed applying modulated differential scanning calorimetry (DSC) method on a DSC-2 installation (Perkin Elmer, Germany) modernized and equipped with the software IFA Gmb (Ulm). The measurements were carried out in a dry air environment in the temperature range from 20 to 330 °C. The heating rate was 2 °С/min.
Results and Discussion
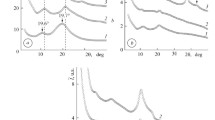
The analysis of wide-angle X-ray diffractograms has shown that IPEC formed of pectin and PEI at a molar ratio of 1:1 is characterized by short-range ordering during translation of fragments of oppositely charged polyelectrolyte macromolecular chains in space. This circumstance is indicated by the appearance of one diffuse diffraction maximum with 2θ m ~20.8° on the X-ray diffractogram of the IPEC sample (Fig. 1, curve 1). The average value of the period of short-range ordering of fragments of complementary macromolecular chains of oppositely charged polyelectrolytes in the IPEC (the Bragg distance between the macromolecule chains of anionic and cationic polyelectrolytes in the IPEC) according to the Bragg equation is
where λ is the wavelength of the characteristic X-ray radiation, which is 4.3 Å (λ = 1.54 Å for СuK α radiation). Once the IMC is formed, the diffraction pattern changes. This is confirmed by the appearance of an intense diffuse diffraction maximum at 2θ m ~11.2° (curve 2) in the presence of a low-intensity amorphous halo, which, unlike that for the initial IPEC, has an angular position at 2θ m ~20.4° (d ~4.4 Å). This diffraction maximum, according to [24], characterizes the existence of interpolyelectrolyte–metal complexes between the central ions (Cu2+) and ligands. Taking into account the angular position of this diffraction peak on the X-ray diffractogram of the IMC, average Bragg distance d between the macromolecule chains of polyelectrolytes coordinated with Cu2+ cations is found to be 7.9 Å.
With the use of the wide-angle X-ray scattering (WAXS) method, it was previously found [21] that the optimum molar ratio BH4 −:Cu2+ for the reduction of Cu2+ cations by sodium borohydride in the IMC bulk followed by the formation of IPEC–Cu/Cu2O nanocomposites is 6. In the X-ray profile of these composites (curve 3), the intense diffraction maximum at 2θ m ~11.2° which is typical for the above polyelectrolyte–metal complexes is absent, unlike the two low-intensity maxima at 2θ m ~35.6° and 40°, confirming formation of Сu2O particles in the IPEC bulk [25]. The X-ray profile additionally displays two intense maxima at 2θ m ~42.8° and 49.6°, which are due to the structure of metallic copper.
Effective size L of Cu/Cu2O nanoparticles crystallites was found through the Scherrer method [26]:
where К is a constant related to the crystallite shape (for an unknown shape, К = 0.9) and β is the angular half-width (the width at half-height) of a diffraction maximum. It was shown that the average value of L is ~4.5 nm (for the calculation, diffraction maxima at 2θ m = 42.8° and 49.6° (curve 3) were used).
In addition, the formation of nanocomposites containing Cu/Cu2O nanoparticles from IMCs is corroborated by the data of transmission electron microscopy (TEM) (Fig. 2). The analysis of TEM images showed that the average size of Cu/Cu2O nanoparticles distributed randomly in the IPEC matrix is 10 nm; variation in sizes is ± 2 nm.
Analyzing WAXS patterns of the initial nanocomposite and that formed under constant electric and magnetic fields, it was found that the chemical reduction of Cu2+ cations in the IMC bulk (at the molar ratio BH4 −:Cu2+ = 6) under constant field results in nanocomposite of the IPEC and metal copper nanoparticles. This outcome is confirmed by the appearance of two diffuse diffraction maxima at 2θ m ~43° and 50° and the absence of two maxima at 2θ m ~35.6° and 40.0°, indicating presence of Сu2O nanoparticles in the IPEC bulk (Fig. 3, curves 1–3).
In parallel, while studying constant electric and magnetic fields’ effect on the structural organization of nanocomposites, the constant fields’ influence on thermomechanical and thermophysical properties of nanocomposites was examined as well.
Analysis of the thermomechanical IPEC’s curve (Fig. 4a, curve 1) demonstrated temperature transitions that are associated with the temperatures of the glass transition and flow occur in the temperature ranges 25–145 °С and 265–350 °С, respectively. Furthermore, in the range of temperatures 150–245 °С, there is a temperature transition that is likely due to the melting of the crystallites of pectin in the IPEC [27]. Respectively, the strong deformational change has been observed in the melting process of pectin’s crystalline phase in IPEC [28]. This is also confirmed by the X-ray diffraction analysis data. Comparing IPEC wide-angle X-ray profiles at Т = 20 ± 2 °С and Т = 170 ± 2 °С (according to the thermomechanical analysis data, the pectin moiety’s melting temperature in the IPEC is 170 ± 2 °С) with profile of pure pectin (powder) (Fig. 4b, curves 1–3), we can conclude that the following processes are taking place successively under the temperature growth IPEC: destruction of interpolyelectolyte complexes and crystallization of pectin fraction. These processes are accompanied by considerable diffraction peak’s displacement towards smaller scattering angels 2θ (from 20.8° to 17.4°), indicating the short-range ordering of IPEC parts—macromolecular chains’ fragments relating to both anion and cation polyelectrolytes, and, thus, resulting in the growth of the Bragg average distance between the macromolecules’ chains from 4.3 to 5.1 Å (Fig. 4b, curves 1, 2). Also, one can see the diffraction maxima at 2θ m = 25.0°, corresponding to pectin’s crystalline structure (Fig. 4b, curves 2).
a Thermomechanical curves of 1 the IPEC, 2 the IMC, and 3 the IPEC–Cu/Cu2O nanocomposite obtained via the chemical reduction of Cu2+ cations in the IMC at a molar ratio BH4 −:Cu2+ = 6. b Wide-angle X-ray diffractograms of IPEC based on pectin and PEI at Т = 20 ± 2 °С (1) and Т = 170 ± 2 °С (2) and pectin (powder) (3). c Wide-angle X-ray diffractograms of IMC at Т = 20 ± 2 °С (1) and Т = 190 ± 2 °С (2). d Thermomechanical curves of nanocomposites obtained via the chemical reduction of Cu2+ cations in the IMC at a molar ratio BH4 −:Cu2+ = 6 in the initial state 1 on the base of the IPEC and Cu/Cu2O and on the base of the IPEC and Cu under 2 constant electric field and 3 constant magnetic field
The formation of IMC leads to the appearance of a temperature transition at T = 205 °С on the thermomechanical curve, which seems to be due to melting of CuSO4 in the IMC bulk [29] that results in the transition of the polymer to the viscous-flow state (Fig. 4a, curve 2). In its turn, Fig. 4c presents IMC profiles fixed at T = 20 ± 2 °С and T = 190 ± 2 °С.
In the IMC diffractogram monitored at T = 190 ± 2 °С, intensive diffraction peak at 2θ m ≈ 11.2° (evidencing the presence of polyelectrolyte–metal complexes) is absent, but new peaks emerge at 2θ m ≈ 43.5 and 50.5° that indicate the existence of copper’ crystalline structure [25]. Therefore, location of the IMC’s thermomechanical curve indicates that in the temperature area ranged from 170 to 205 °С (see Fig. 4a, curve 2), the following successive processes occur in IMC sample: interpolyelectrolyte–metal complexes are destroyed, and then, the salt (CuSO4) transfers from its ionic form to the crystalline one and then melts.
Analysis of the thermomechanical curves of the IPEC, the IMC, and IPEC–Cu/Cu2O nanocomposite (see Fig. 4a) shows that during the transition from the IPEC to the IMC, glass-transition temperature Т g increases, and while IMC is converting into the IPEC–Cu/Cu2O nanocomposite, Т g significantly decreases (Table 1).
Simultaneously, while T g is changed, the decrease of transition temperature to the viscous-flow state (T f) occurs in the following row:
Relative deformation value of polymer systems is seen in Table 1.
In addition, observing thermomechanical curves of the nanocomposites filled with Cu/Сu2O nanoparticles prepared without subjection to constant field and the IPEC–Cu nanocomposites formed under the influence of constant electric and magnetic fields (Fig. 4d), one can see that Т g and T f values are increased for the IPEC–Cu nanocomposite formed under electric and magnetic fields. This result indicates the higher thermal stability of this polymer system compared with its analog obtained without subjection to a field. At the same time, in nanocomposites affected by magnetic and electrical fields, anionic polyelectrolyte pectin’s melting of crystalline phase takes place in a wide temperature range between 170 and 250 °С, when nanocomposites without any field’s action reveal this interval at 180–230 °С (Fig. 4d). This effect is supposed to be due to formation more ordered structure in polymer matrix, when composites formed under impact of a physical field.
Analysis of the IPEC (pectin–PEI), IMC (pectin–Cu2+–PEI), and nanocomposites IPEC–Cu/Cu2O thermograms revealed that their transition temperatures are in a good agreement with those seen in the thermomechanical curves of these polymer systems (see Fig. 5a and Fig. 4a).
a Thermograms of 1 the IPEC, 2 the IMC, and 3 the IPEC–Cu/Cu2O nanocomposite obtained via the chemical reduction of Cu2+ cations in the IMC at a molar ratio BH4 −:Cu2+ = 6. b Thermograms of nanocomposites obtained via the chemical reduction of Cu2+ cations in the IMC at a molar ratio BH4 −:Cu2+ = 6 in the initial state 1 on the base of the IPEC and Cu/Cu2O and on the base of the IPEC and Cu under 2 constant electric field and 3 constant magnetic field
For IPEC at T = 185 °С and IMC at T = 202 °С, there are maximums, connected with pectin melting and CuSO4, correspondingly. Also, the minimums observed at T = 203 °С for IPEC and T = 218 °С for IMC could be explained by destructive processes proceeded in these systems (Fig. 5a, curves 1, 2). In nanocomposite’s thermogram, there exist two maximums at T = 147 and 208 °С, corresponding to pectin’s crystallites melting, which have lower and higher melting temperatures, respectively (Fig. 5a, curves 3).
In its turn, in thermograms of nanocomposites based on IPEC and Cu/Cu2O nanoparticles which were formed in original state (without any field) and those, having IPEC and Cu nanoparticles only, being formed under the action of constant electric and magnetic fields (Fig. 5b), one can observe that latter is characterized by higher Т g and ∆Т g values compared to systems prepared in the initial state (when a field is absent) (Table 2).
Conclusions
Constant electric and magnetic fields impact the structural organization and thermomechanical and thermophysical properties of nanocomposites based on the pectin–polyethyleneimine interpolyelectrolyte complex, and Cu nanoparticles formed from pectin–Cu2+–polyethyleneimine interpolyelectrolyte–metal complexes, involving sodium boron hydride as reducing agent, have been studied. It was found that the chemical reduction of Cu2+ cations in the IMC bulk under constant field proceeds with the formation of IPEC-based nanocomposite and nanoparticles of just metal Cu phase, whereas IPEC–Cu/Cu2O nanocomposite is formed in the absence of field.
Thermomechanical and thermophysical analyses reveal considerable changes in glass-transition temperatures in the following row—interpolyelectrolyte complex—interpolyelectrolyte–metal complex and nanocomposites based on interpolyelectrolyte complex–Cu/Cu2O. It has been shown that, under constant field, nanocomposites obtained on the base of interpolyelectrolyte complex–Cu have the higher structural glass-transition temperatures and thermal stability.
References
Gates BC, Guezi L, Knosinger H (1986) Metal clusters in catalysis. Elsevier, Amsterdam
Pomogailo AD, Rozenberg AS, Uflyand IE (2000) Metal nanoparticles in polymers. Khimiya, Moscow, in Russian
Nicolais L (2005) Metal-polymer nanocomposites. Wiley, New York
Schubert US, Newkome GR, Manners I (2006) Metal-containing and metallosupramolecular polymers and materials. ACS, Washington
Wohrle D, Pomogailo AD (2003) Metal complexes and metals in macromolecules: synthesis, structure and properties. Wiley-VCH, Weinheim
Pomogailo AD, Kestelman VN (2005) Metallopolymer nanocomposites. Springer, New York
Yan Y, Huang J (2010) Hierarchical assemblies of coordination supramolecules. Coord Chem Rev 254:1072–1080
Zezin AB, Rogacheva VB, Feldman VI, Afanasiev P, Zezin AA (2010) From triple interpolyelectrolyte-metal complexes to polymer-metal nanocomposites. Adv Colloid Interface Sci 158:84–93
Bruening ML, Dotzauer DM, Jain P, Ouyang L, Baker GL (2008) Creation of functional membranes using polyelectrolyte multilayers and polymer brushes. Langmuir 24:7663–7673
Liu G, Dotzauer DM, Bruening ML (2010) Ion-exchange membranes prepared using layer-by-layer polyelectrolyte deposition. J Membr Sci 354:198–205
Ruiz P, Macanas J, Munoz M, Muraviev DN (2011) Intermatrix synthesis: easy technique permitting preparation of polymer-stabilized nanoparticles with desired composition and structure. Nanoscale Res Lett 6:343
Macanas J, Ouyang L, Bruening ML, Munoz M, Remigya J-C, Lahittea J-F (2010) Development of polymeric hollow fiber membranes containing catalytic metal nanoparticles. Catal Today 156:181–186
Nicolais L (2005) Carotenuto G ed: metal-polymer nanocomposites. Wiley-VCH, Weinheim
Shih C-M, Shieh Y-T, Twu Y-K (2009) Preparation of gold nanopowders and nanoparticles using chitosan suspensions. Carbohydr Polym 78:309–315
Geng B, Jin Z, Li T, Qi X (2009) Preparation of chitosan-stabilized Fe0 nanoparticles for removal of hexavalent chromium in water. Sci Total Environ 407:4994–5000
Yoksan R, Chirachanchai S (2009) Silver nanoparticles dispersing in chitosan solution: preparation by γ-ray irradiation and their antimicrobial activities. Mater Chem Phys 115:296–302
Long D, Wu G, Chen S (2007) Preparation of oligochitosan stabilized silver nanoparticles by gamma irradiation. Radiat Phys Chem 76:1126–1131
Sergeev GB (2003) Nanochemistry. MGU, Moscow, in Russian
Zezin AB, Rogacheva VB, Valueva SP, Nikonorova NI, Zantsokhova MF, Zezin AA (2006) From triple interpolyelectrolyte-metal complexes to polymer-metal nanocomposites. Nanotechnol Ross 1:191–200 (in Russian)
Ostaeva GY, Selishcheva ED, Pauton VD, Papisov IM (2008) Pseudotemplate synthesis of copper nanoparticles in solutions of poly(acrylic acid)–pluronic blends. Vysokomol Soedin 50:1102–1106 (in Russian)
Demchenko VL, Shtompel VI, Riabov SV, Goncharenko LA (2014) Structure, thermomechanical and electrical properties of nanocomposites formed from triple polyelectrolyte-metal complexes pectin–Cu(ΙΙ)–polyethyleneimine. Material Science of Nanostructures 1:45–52 (in Ukrainian)
Demchenko VL, Shtompel VI, Goncharenko LA, Kobylinskiy SM, Riabov SV (2015) The influence of a constant magnetic field on the structure and thermomechanical properties of polymer-metal systems based on polyelectrolyte complexes of pectin and polyethyleneimine with copper ions and nanocomposites formed from these systems. Polym J 37:144–150, in Ukrainian
Kobylins’kii SM, Riabov SV, Kercha YY (2005) Chitosan modification by polyethyleneimines. Vopr Khim Khim Tekhnol 5:28–33 (in Ukrainian)
Shtompel’ VI, Kercha YY (2008) Structure of linear polyurethanes. Naukova dumka, Kiev, in Russian
Kou T, Jin C, Zhang C, Sun J, Zhang Z (2012) Nanoporous core–shell Cu@Cu2O nanocomposites with superior photocatalytic properties towards the degradation of methyl orange. RSC Adv 2:12636–12643
Gin’e A (1961) X-ray diffraction of crystals. Theory and practice. Fizmatgiz, Moscow, in Russian
Donchenko LV, Firsov GG (2007) Pectin: main properties, production and application. DeLi print, Moscow, in Russian
Teitelbaum BJ (1979) Thermomechanical analysis of polymers. Nauka, Moscow, in Russian
Lidin RA, Molochko VA, Andreeva LL (1996) The chemical properties of inorganic substances. Khimiya, Moscow, in Russian
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Demchenko, V., Shtompel’, V., Riabov, S. et al. Constant Electric and Magnetic Fields Effect on the Structuring and Thermomechanical and Thermophysical Properties of Nanocomposites Formed from Pectin–Cu2+–Polyethyleneimine Interpolyelectrolyte–Metal Complexes. Nanoscale Res Lett 10, 479 (2015). https://doi.org/10.1186/s11671-015-1181-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s11671-015-1181-z