Abstract
Single-phase mixed aluminates-titanates Pr1−x Sr x Al1−x Ti x O3 (x = 0.1, 0.2, 0.3, 0.5, 0.7) with rhombohedral perovskite structure were prepared by solid-state reaction technique at 1823–1873 K. Morphotropic rhombohedral-to-cubic phase transition in Pr1−x Sr x Al1−x Ti x O3 series is predicted to occur at x = 0.88. The temperature-induced structural phase transition R \( \overline{3} \) с − Pm \( \overline{3} \) m in Pr0.5Sr0.5Al0.5Ti0.5O3, detected at 930 K by in situ high-temperature X-ray synchrotron powder diffraction, occurs at considerably lower temperature as the corresponding transformation in the parent compound PrAlO3 (1770 K). Such remarkable drop of the transition temperature is explained by gradual decrease of the perovskite structure deformation in the Pr1−x Sr x Al1−x Ti x O3 series with increasing Sr and Ti contents as a consequence of the increasing Goldschmidt tolerance factor. For Pr0.3Sr0.7Al0.3Ti0.7O3 phase, a sequence of the low-temperature phase transformation R \( \overline{3} \) с − Immb(C2/m) − I4/mcm was detected.
Similar content being viewed by others
Background
Rare earth aluminates RAlO3 with perovskite structure and SrTiO3-based materials show diverse technological application. In particular, they are used as solid electrolytes and anode materials in solid oxide fuel cells, as substrates for thin film epitaxy, materials for laser hosts, scintillates and phosphors, high-temperature ceramics and refractory materials ([1–5] and references herein). Due to the opposite signs of the temperature coefficient of the resonant frequency (τ f ) of the RAlO3 and SrTiO3 compounds, mixed aluminates-titanates formed in the RAlO3–SrTiO3 systems are considered as prospective microwave materials with a high dielectric constant, moderate quality factor and a near zero value of τ f [5–7].
During the last decade, RAlO3–SrTiO3 systems are of considerable interest in the physics of materials used in modern engineering. The two-dimensional electron gas at the interface between two insulators LaAlO3 and SrTiO3 [8] has been an active research area in the field-tunable metal-insulator transition, 2D superconductivity, coexistence of superconductivity and ferromagnetism, etc. [9–11]. Just recently, a similar effect was reported on the interfaces of SrTiO3 and RAlO3 (R = La, Pr, Nd) and RGaO3 compounds (R = La, Nd) in both crystalline and amorphous forms [12].
The aim of the present work is the study of the phase and structural behaviour of the mixed aluminates-titanates formed in the PrAlO3–SrTiO3 pseudo-binary system. At room temperature, the end members of the system—PrAlO3 and SrTiO3—adopt different variants of perovskite structure—rhombohedral R \( \overline{3} \) с and cubic Pm \( \overline{3} \) m, respectively. Rhombohedral PrAlO3 transforms into the cubic perovskite structure at about 1770 K ([4], and references herein). In addition, PrAlO3 undergoes a sequence of low-temperature (LT) structural phase transformations from the rhombohedral to an orthorhombic Imma structure at 205 K and from orthorhombic to a monoclinic C2/m structure at 151 K ([4], and references herein). Strontium titanate SrTiO3 undergoes a low-temperature structural phase transition from the cubic to the tetragonal I4/mcm perovskite structure below 105 K [13, 14]. Owing to the abovementioned peculiarities of the crystal structures PrAlO3 and SrTiO3 and their structural instabilities, extremely complex phase and structural behaviour are expected in the mixed praseodymium-strontium aluminate-titanate system.
Methods
Mixed aluminates-titanates Pr1−x Sr x Al1−x Ti x O3 (x = 0.1, 0.2, 0.3, 0.5, 0.7) were prepared from stoichiometric amounts of the constituent oxides Pr6O11, Al2O3, TiO2 and strontium carbonate SrCO3 by solid-state reaction technique. The precursor powders were ball milled in ethanol for 3–6 h, dried, pressed in the pellets and sintered in air at 1673–1773 K for 18 h (the samples with x = 0.1 and 0.2) and at 1593 K for 24 h (the samples with x = 0.3, 0.5 and 0.7). After regrinding and powdering, the obtained products were pressed in the pellets and repeatedly fired in air at 1873 K (x = 0.1 and 0.2) and 1823 K (x = 0.3, 0.5 and 0.7) for 10 h.
X-ray powder diffraction technique (Huber imaging plate Guinier camera G670, Cu Kα1 radiation) was used for the phase and structural characterization of the samples at room temperature. Thermal behaviour of the mixed aluminates-titanates has been studied exemplarily on Pr0.5Sr0.5Al0.5Ti0.5O3 and Pr0.3Sr0.7Al0.3Ti0.7O3 samples in the temperature ranges of 298–1173 K and 20–298 K, respectively. Corresponding in situ high-resolution X-ray synchrotron powder diffraction experiments were performed at beamlines B2 at HASYLAB/DESY (Hamburg, Germany) and ID22 at ESRF (Grenoble, France) during beamtimes allocated to the experiments I-20110214 and hc2044, respectively.
All crystallographic calculations including full-profile Rietveld refinement were performed by using WinCSD program package [15].
Results and Discussion
Examination of X-ray powder diffraction patterns revealed a formation of the single-phase perovskite structures in all samples synthesised (Fig. 1). No traces of parasitic phases were detected. An analysis of the splitting of diffraction maxima in the Pr1−x Sr x Al1−x Ti x O3 patterns with x = 0.1, 0.2, 0.3 and 0.5 proves rhombohedral deformation of the perovskite structure. The rhombohedral splitting decreases with the increase in the content of strontium and titanium in the Pr1−x Sr x Al1−x Ti x O3 series, completely vanishing in the Pr0.3Sr0.7Al0.3Ti0.7O3 sample. However, the presence of a weak (113) reflection at the diffraction pattern of this sample (Fig. 1) indicates that its structure still remains rhombohedral.
From the experimental XRD patterns of Pr1−x Sr x Al1−x Ti x O3 samples, the crystal structure parameters of the mixed aluminates-titanates were derived. Full-profile Rietveld refinement performed in space group R \( \overline{3} \) с resulted in excellent agreement between calculated and experimental profiles (see, for example, Fig. 2) and led to the final structural parameters and residuals presented in Table 1.
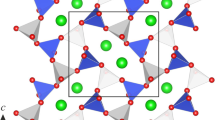
Graphical results of Rietveld refinement of Pr0.8Sr0.2Al0.8Ti0.2O3. The experimental XRD pattern (blue dots) is shown in comparison with the calculated one. Vertical bars indicate positions of diffraction maxima in space group R \( \overline{3} \) с. The inset shows the view of the structure as corner-shared Al/TiO6 octahedra with Pr/Sr species located between them
Comparison of the obtained structural parameters of the praseodymium-strontium mixed aluminates-titanates with the literature data for the “pure” PrAlO3 and SrTiO3 (Fig. 3) clearly proves the formation of the extended solid solution Pr1−x Sr x Al1−x Ti x O3 with rhombohedral perovskite structure. A morphotropic phase transition from rhombohedral to the cubic perovskite structure in the Pr1−x Sr x Al1−x Ti x O3 series can be expected at x = 0.88, as it follows from the analysis of the concentration dependence of the unit cell dimensions of the rhombohedral lattice (Fig. 3). In the related LaAlO3–SrTiO3 system, the rhombohedral solid solution exists up to 60 mole % of LaAlO3; after that, the transition to the cubic perovskite structure takes place [16].
In situ high-temperature X-ray synchrotron powder diffraction investigation of the Pr0.5Sr0.5Al0.5Ti0.5O3 sample revealed a continuous phase transition from rhombohedral to the cubic perovskite structure at elevated temperatures. As it was established from the temperature-resolved X-ray synchrotron powder diffraction measurements, the rhombohedral lattice parameters a and c increase anisotropically with temperature and merge together at 930 K, when the transition to the ideal perovskite structure occurs (Fig. 4).
In comparison with the parent PrAlO3 compound, in which transformation to the cubic perovskite structure occurs around 1770 K [4], the R \( \overline{3} \) с − Pm \( \overline{3} \) m transition in Pr0.5Sr0.5Al0.5Ti0.5O3 takes place at considerably lower temperature of 930 K. Such remarkable drop of the phase transition temperature can be explained by gradual decrease of the perovskite structure deformation in the Pr1−x Sr x Al1−x Ti x O3 series with increasing of Sr and Ti content. According to the phase diagram of the RAlO3-based perovskite systems [4], the temperature of the R \( \overline{3} \) с − Pm \( \overline{3} \) m phase transition decreases linearly with increasing radii of R cation as a consequence of the increasing Goldschmidt tolerance factor.
Graphical results of Rietveld refinement of the high-temperature modifications of the Pr0.5Sr0.5Al0.5Ti0.5O3 structure and refined structural parameters at selected temperatures are presented in Fig. 5 and Table 2, respectively.
X-ray synchrotron powder diffraction patterns of Pr0.5Sr0.5Al0.5Ti0.5O3 (HASYLAB B2 data, λ = 0.53820 Å). Full-profile Rietveld refinement was performed in space groups Pm \( \overline{3} \) m (T = 1173 K) and R \( \overline{3} \) с (T = 373 K). Experimental (dots) and calculated patterns, difference profiles and positions of the diffraction maxima are given
Spot-check examination of low-temperature structural behaviour of the Pr1−x Sr x Al1−x Ti x O3 system was performed on the example of a Pr0.3Sr0.7Al0.3Ti0.3O3 sample at temperatures of 20, 80, 160 and 220 K. Extremely high resolution of the beamline ID22 at ESRF allows to detect subtle changes in the reflections splitting at different temperature measurements (Fig. 6), which clearly prove a sequence of LT phase transformations in this sample.
Crystal structures of Pr0.3Sr0.7Al0.3Ti0.3O3 at RT and at 220 K were refined in the space group R \( \overline{3} \) с, thus confirming the results derived from the convenient XRD data (Table 1). Taking into account the character of the reflection splitting, crystal structure parameters of the low-temperature modification of Pr0.3Sr0.7Al0.3Ti0.3O3 at 20 and 80 K were successfully refined in space group I4/mcm. X-ray synchrotron diffraction features of Pr0.3Sr0.7Al0.3Ti0.3O3 at 160 K could be successfully modelled either in the orthorhombic Immb or in the monoclinic I2/m (C2/m) perovskite structure. Since in both cases during the refinement procedures, practically the same residuals were obtained, a preference should be given to the more symmetric orthorhombic structure. Taking into account that the end members of the system show different sequences of LT phase transformations R \( \overline{3} \) с − Immb − C2/m (PrAlO3) and Pm \( \overline{3} \) m − I4/mcm (SrTiO3), additional investigations are required in order to shade light on the complex phase and structural behaviour of the mixed aluminates-titanates below RT.
Conclusions
The formation of extended solid solution Pr1−x Sr x Al1−x Ti x O3 with rhombohedral perovskite structure has been revealed in the PrAlO3–SrTiO3 pseudo-binary system based on X-ray powder diffraction data. An analysis of the obtained structural parameters in comparison with the data for the parent compounds PrAlO3 and SrTiO3 revealed a decrease of perovskite structure deformation in Pr1−x Sr x Al1−x Ti x O3 series with increasing Sr and Ti content as a consequence of the increasing Goldschmidt tolerance factor. As a result, concentration-induced phase transition from a rhombohedral to the cubic perovskite structure takes place in the Pr1−x Sr x Al1−x Ti x O3 system at x = 0.88. A decreasing structure deformation in Pr1−x Sr x Al1−x Ti x O3 series leads to the significant decrease of the temperature-induced phase transition R \( \overline{3} \) с − Pm \( \overline{3} \) m from 1770 to 930 K in PrAlO3 and Pr0.5Sr0.5Al0.5Ti0.5O3, respectively. The sequence of the low-temperature phase transition R \( \overline{3} \) с − Immb(I2/m) − I4/mcm in Pr0.3Sr0.7Al0.3Ti0.3O3 revealed during the spot-check X-ray synchrotron powder diffraction examination promises an extremely complex picture of the phase and structural relations in PrAlO3–SrTiO3 system below room temperature. Further structural and calorimetric investigations of the system are in progress.
References
Lybye D, Poulsen FW, Mogensen M (2000) Conductivity of a- and b-site doped LaAlO3, LaGaO3, LaScO3 and LaInO3 perovskites. Solid State Ionics 128:91–103
Takamura H, Enomoto K, Aizumi Y, Kamegawa A, Okada M (2004) Preparation and oxygen permeability of Pr–Al-based perovskite-type oxides. Solid State Ionics 175:379–382
Inagaki Y, Suzuki S, Kagomiya I, Kakimoto K, Ohsato H, Sasaki K, Kuroda K, Shimada T (2007) Crystal structure and microwave dielectric properties of SrTiO3 doped LaAlO3 single crystal grown by FZ. J Eur Ceram Soc 27:2861–2864
Vasylechko L, Senyshyn A, Bismayer U (2009) Perovskite-type aluminates and gallates. In: Gschneidner KA Jr, Bünzli J-CG, Pecharsky VK (eds) Handbook on the physics and chemistry of rare earths, vol 39. North-Holland, Netherlands, pp 113–295
Yaremchenko AA, Patrício SG, Frade JR (2014) Thermochemical behavior and transport properties of Pr-substituted SrTiO3 as potential solid oxide fuel cell anode. J Power Sources 245:557–569
Verbraeken MC, Ramos T, Agersted K, Ma Q, Savaniu CD, Sudireddy BR, Irvine JTS, Holtappels P, Tietz F (2015) Modified strontium titanates: from defect chemistry to SOFC anodes. RSC Adv 5:1168–1180
Qu J, Yuan Ch, Liu F, Liu X, Chen G, Qin P. Microstructures and microwave dielectric properties of (1−x)Sr0.2Na0.4Sm0.4TiO3−x LnAlO3 (Ln = Nd, Pr and Sm) ceramic systems. J Mater. Sci.: Mater Electronics. 2015; 26: 4862–4869
Ohtomo A, Hwang HY (2004) A high-mobility electron gas at the LaAlO3/SrTiO3 heterointerface. Nature 427:423–426
Thiel S, Hammerl G, Schmehl A, Schneider CW, Mannhart J (2006) Tunable quasi-two-dimensional electron gases in oxide heterostructures. Science 313:1942–1945
Reyren N, Thiel S, Caviglia AD, Fitting Kourkoutis L, Hammerl G, Richter C, Schneider CW, Kopp T, Rüetschi A-S, Jaccard D, Gabay M, Muller DA, Triscone J-M, Mannhart J (2007) Superconducting interfaces between insulating oxides. Science 317:1196–1199
Dikin DA, Mehta M, Bark CW, Folkman CM, Eom CB, Chandrasekhar V (2011) Coexistence of superconductivity and ferromagnetism in two dimensions. Phys Rev Lett 107:056802
Li C, Liu Z, Lü W, Wang XR, Annadi A, Huang Zh, Zeng Sh, Ariando, Venkatesan T. Tailoring the two dimensional electron gas at polar ABO3/SrTiO3 interfaces for oxide electronics. Scientific Reports. 2015; doi: 10.1038/srep13314
Kiat JM, Roisnel T (1996) Rietveld analysis of strontium titanate in the Müller state. J Phys Condens Matter 8:3471–3415
Hayward SA, Salje EKH (1999) Cubic-tetragonal phase transition in SrTiO3 revisited: Landau theory and transition mechanism. Phase Transit 68(3):501–522
Akselrud L, Grin Y (2014) WinCSD: software package for crystallographic calculations (version 4). J Appl Crystallogr 47:803–805
Cho SY, Hong KS, Ko K-H (1999) Mixture-like behavior in the microwave dielectric properties of the (1–x)LaAlO3–x SrTiO3 system. Mater Res Bull 34(4):511–516
Acknowledgements
The work was supported in part by the Ukrainian Ministry of Education and Sciences (Project “KMON”) and ICDD Grant-in-Aid program. The authors thank A. Fitch for kind assistance with high-resolution synchrotron powder diffraction measurements at ESRF during beamtime allocated to the experiment hc2044.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing Interests
The authors declare that they have no competing interests.
Authors’ Contributions
LV synthesised the samples, performed the HT synchrotron powder diffraction measurements at HASYLAB and structural characterization of the samples and wrote the manuscript. RS contributed to the data evaluation, figure and table preparation and manuscript writing. YP and HR performed the laboratory X-ray and LT synchrotron diffraction measurements at ESRF. All authors read and approved the final manuscript.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Vasylechko, L., Stepchuk, R., Prots, Y. et al. Concentration- and Temperature-Induced Phase Transitions in PrAlO3–SrTiO3 System. Nanoscale Res Lett 11, 17 (2016). https://doi.org/10.1186/s11671-015-1225-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s11671-015-1225-4