Abstract
The m-aminophenylarsonic acid (m-APAA) was immobilized onto the silica gel surface with covalently grafted quaternary ammonium groups via ion exchange. The diazotization of ion-bonded m-APAA resulted in a new solid-phase spectrophotometric reagent for detection of 1-naphtol in environmental water samples. The procedure of solid-phase spectrophotometric analysis is characterized by 20 μg L−1 limit of detection (LOD) of 1-naphtol, up to 2000 concentration factor, and insensitivity to the presence of natural water components as well as to 30-fold excess of phenol, resorcinol, and catechol.
Similar content being viewed by others
Background
Highly toxic and mutagenic derivatives of naphthols and aminonaphthols constitute an important class of environmental pollutants [1, 2]. Most of the naphthols appear in the environmental water due to the biodegradation of pesticides and azo dyes [3, 4]. For example, environmental decomposition of carbaryl (1-naphthyl-n-methylcarbamate), which is widely used as an insecticide, gives 1-naphthol [3], whereas common azo dyes give harmful pollutant amino-2-naphthol [4].
Usually, the naphthols are analyzed by high-performance liquid chromatography [5], flow-injection analysis with further photometric detection [6], fluorimetry [7–9], phosphorimetry [10], and by immunosensors [11]. Fluorimetric, phosphorimetric, and immunosensor methods require complicated sample pre-treatments. Photometric method is much more reliable for infield analysis; however, its sensitivity is insufficient for most of the environmental cases [9]. Therefore, the pre-concentration of naphthol derivates by means of liquid-liquid or solid-phase extraction (SPE) is commonly used in sample preparation prior to photometric determination [9, 10, 12–14]. Several approaches for selective SPE were proposed in literature: (1) pre-concentration of 1-naphthol derivate on silica with immobilized Co3+ [12] or polyurethane foam [13]; (2) formation of inclusion complexes of naphthols and 3-Br-1-propanol in β-cyclodextrine [10]; (3) microextraction of 1-naphthol on a glass capillary modified with polydimethylsiloxane-divinylbenzene [14].
The selectivity of pre-concentration procedure appeared the major concern for environmental samples analysis. Developments in this area are still needed due to only few reports on selective adsorbents preparation [15–17]. It is even more challenging to develop selective solid-phase reagent allowing the naked-eye monitoring of naphthol contaminations in environmental water.
The reaction of azo coupling, taking place between diazonium salts (DS) and aromatic amines, phenols, or naphthols, results in deep-colored products (azo dyes). Therefore, it is widely used for photometric analysis of organic pollutants [18–20]. However, due to modest selectivity and insufficient sensitivity, the azo coupling is not fully applicable on environmental samples. This limitation can be overcome if the procedures of pre-concentration and analysis are combined in one solid-phase analytical reagent (SPAR). To develop such a SPAR, we propose immobilization of azo coupling reagent on the silica gel surface.
The present work aimed to develop a new SPAR for selective pre-concentration of 1-naphthol traces from water samples. SPAR changes its color, with no additional reagent added, as the pollutant’s concentration increases up to its maximum allowed concentration (MAC). To evaluate the level of the pollutant in a water sample, a UV–vis diffuse reflection spectroscopy can be used. The SPAR, which is introduced in this work, is based on m-aminophenylarsonic acid (m-APAA), immobilized on silica with grafted quaternary ammonium anion exchanging groups. The azo coupling between 1-naphthol and the DS, formed by diazotation of immobilized m-APAA, ensures high efficiency of the pollutant pre-concentration and selectivity of SPE; intense color of azo product means low detection limit.
Methods
Apparatus
Measurements of pH and electrode potentials were performed by a laboratory ion-meter I-160M (Antech, Belarus). A peristaltic pump 2132 LKB Bromma was used to set liquid flow rates in dynamic adsorption experiments. The UV–vis absorbance spectra of solutions were measured by an UV-2401 PC (Shimadzu) spectrophotometer in 220–900-nm range; the diffuse reflection UV–vis (DR-UV) spectra of solids were recorded by a CS-9301 PC densitometer (Shimadzu).
Reagents and Materials
Reagent grade chemicals (Merck) were used without further purification. m-Aminophenylarsonic acid was obtained from m-nitrophenylarsonic acid by its reduction with ferrous sulfate in water solution according to the literature method [21]. The рН values of solutions were maintained by buffer prepared from 0.05 mol∙L−1 sodium tetraborate and hydrochloric acid. The concentration of m-APAA and its diazotized product was measured by spectrophotometry according to [22]. Concentrations of 1-naphthol, resorcinol, and catechol were measured by spectrophotometry after the reaction with 4-aminoantipyrin according to [23].
Synthesis of Trimethyl(3-Trimethoxysilylpropyl)Ammonium Iodide (TMPA)
Syntheses of TMPA and silica with covalently immobilized TMPA (SiO2–TMPA) were performed in anhydrous solvents under Ar. The TMPA was obtained according to Hoffman reaction (Scheme 1).
For this, 0.01 mol of 3-aminopropyltrimethoxysilane was mixed with 0.04 mol of methyl iodide in 50 mL of methanol at room temperature. After during 1 h time 0.02 mol of CH3ONa in 10 mL of CH3OH was added dropwise to the reaction mixture followed by stirring for 20 h. After the solvent was evaporated in vacuum (0.1 mmHg), white residual, giving 0.4 t(2H), 1.65–1.8 m(2H), 3.1–3.3 m(11H), 3.51 s(9H) ppm signals in 1H NMR spectrum in CDCl3 solution, was used in SiO2–TMPA with no further purification.
Synthesis of Silica with Grafted Anion Exchange Groups (SiO2–TMPA)
Prior functionalisation, silica gel carrier was annealed for 8 h in air at 500 °C. Activated on such way, silica gel (8 g) was immersed to 50 mL of CH3CN and TMPA was added. The suspension was stirred during 15 h at 80 °C and then the silica was filtered, washed with CH3CN in the Soxhlet extractor for 5 h, and dried in vacuum. Obtained organo-silica was immersed to saturated KBr aqueous solution for 5 min, washed with this water by decantation, and finally dried on air at 105 °С. The concentration of alkylammonium groups immobilized on SiO2–TMPA was determined from argentometric titration as 552 μmol g−1.
Synthesis of Silica with Immobilized m-АPAA (SiO2–m-АPAA)
Aqueous solution of m-АPAA (25 mL, 1.2 × 10−2 mol L−1) was passed through the column (d = 5 mm, h = 100 mm) filled with 0.5 g of SiO2–TMABr with 0.5 mL min−1 flow rate. Obtained SiO2–m-АPAA was washed with distilled water and dried at room temperature for 24 h. The concentration of immobilized m-АPAA groups was calculated as a difference between the concentration of m-АPAA in solution before and after passing through the column and the concentration found in the distilled water.
Synthesis of Silica Modified with m-Diazophenylarsonic Acid (SiO2–DS) and its Hydrolytic Stability
Diazotization of immobilized m-APAA was performed according to reported procedure [22]. For this, 50 mg of SiO2–m-АPAA was mixed with 5 mL of 5 × 10−2 mol L−1 NaNO2 and 5 mL of 0.5 mol L−1 HCl. The mixture was shook for a while. The adsorbent was washed quickly with distilled water followed by buffer solution (pH = 8). Since the diazonium salts are thermally unstable, all procedures were carried out below 5 °C.
Adsorption Studies of SiO2–DS
The adsorption capacity of SiO2–DS to phenolic compounds was determined in dynamic conditions. Aqueous solutions of phenols (5 × 10−4 mol L−1) having pH 8 were passed (0.2 mL min−1) through a column packed with 300 mg of SiO2–DS. The concentration of phenols was determined in each 2 mL portion of the effluent. The adsorption capacity of SiO2–DS was calculated by Eq. (1):
where k is the number of solution portions passed through the column, g is the mass of adsorbent in the column, C 0 is the initial concentration of phenolic compound in the solution, C i is its concentration in each portion of the effluent, and V is the volume of effluent portion.
The adsorption kinetics was studied in static conditions. Fifty milligrams of SiO2–DS was mixed with 25 mL of 2 × 10−5 mol L−1 solution of the phenolic compound at pH 8 and shaken during fixed time interval (1–10 min). The solid phase was filtered, washed with pH 8 buffer, and dried at room temperature. The solution was analyzed for phenol contents while the solid phase was used to record the UV–vis spectrum.
Solid-Phase Spectrophotometric Detection of 1-Naphthol
To make a calibration curve, portions of aqueous 1-naphthol solutions (95 mL) with a concentration in a range 0–2 mg L−1 were mixed with 5 mL portions of buffer solution (pH = 8). After cooling down into the ice bath, these solutions were added to as-prepared 50 mg portions of SiO2–DS and vigorously stirred for 5 min. The solid phase was filtered, washed with water, and dried at room temperature. The DR-UV spectra were recorded and presented as Kubelka-Munk function (F(R) = (1 − R)2/2R) giving the intensity of spectrum signals which is directly proportional to concentration of absorbing species in the solid phase.
To analyze natural water samples, 95 mL samples (as received or spiked with 100 μg L−1 of 1-naphthol) without any pretreatment were mixed with 5 mL portions of buffer (pH = 8) and treated as described above.
Results and Discussion
Several strategies of the DS immobilization on the silica gel surface are possible: covalent grafting [24, 25], physical adsorption, and ion exchange binding. Due to low stability of diazonium salts in solution and even in immobilized state [26], it is desirable to have fast and simple procedure for the SiO2–DS preparation, which is looking problematically for covalent grafting. The physisorption of organic reagents also seems doubtful, particularly due to desorption of DS at the stage of adsorbent application. To avoid the above drawbacks, the ion exchange approach is likely to be helpful; furthermore, silica-based anion and cation exchangers are thoroughly studied [27] and some of them are commercially available.
Due to the instability of diazonium salts in solution as well as in the immobilized state, a two-stage immobilization approach was used for preparation of SiO2–DS. At the first stage, a stable precursor of diazonium salt (m-APAA) was immobilized onto the surface of silica with covalently grafted quaternary ammonium groups (Scheme 1). To obtain the SiO2–DS, amino groups of immobilized m-APAA were diazotized by treatment with a standard diazotizing mixture (HCl + NaNO2, see Scheme 2).
Synthesis of SiO2–APAA
The m-АPAA molecule contains acidic groups of intermediate strength (−AsO3H2) and weakly basic groups (−NH2). That is why it can exist in aqueous solutions as cationic (H3A+), neutral (H2A), and two anionic (HA− and A2−) forms depending on the pH. Unfortunately, no exact values of the acidity constants were found in the literature for m-APAA. However, the isoionic pH values (i.e., pH i = (pK1 + pK2)/2 which corresponds to maximum concentration of H2A and equal concentrations of H3A+ and HA−) are equal to 3.00 for ortho- and to 3.15 for para-APAA isomers, respectively [28]. These values allow us to assume that in aqueous solutions m-APAA can be either in its cationic H3A+ (pH ≤ 2) or anionic form (pH ≥ 4). Difference in the UV–vis absorption spectra of the m-APAA solutions at different pH values (Fig. 1) is probably caused by deprotonation of the amino group (formation of the auxochrome results in a spectral maximum redshift), which confirms ionization of the m-APAA at pH ≥ 4.
As the silica carrier is not stable at the pH >9 [29] and the m-АPAA exists in a protonated form at pH < 2, the ion exchange immobilization of m-АPAA on the SiO2–TMABr was studied in the pH range of 2–9. Maximum adsorption capacity to m-APAA (С L) is equal to 520 ± 10 μmol/g at pH = 4.0. This value corresponds to more than 94 % conversion of surface bromide salt (С L = 550 ± 10 μmol/g according to argentometric titration data) to m-АPAA salt. Lower adsorption capacities found at other studied pH values may be explained by incomplete deprotonation of m-APAA at lower pH and the presence of doubly charged m-APAA anions at higher pH. That is why the SiO2-APAA for all further experiments was prepared at pH = 4. The position of absorption maximum in the DR-UV spectrum of SiO2–APAA (300 nm, see Fig. 1) coincides with the position of maximum in the solution UV–vis spectra of m-APAA anionic forms. This fact additionally confirms that the ion exchange immobilization of m-APAA on SiO2–TMABr occurs according to Scheme 1.
Synthesis and Properties of SiO2–DS
The SiO2 –DS was obtained by a reaction of SiO2–APAA with NaNO2 in acidic media (Scheme 2). However, the diazonium salt tends to desorb in these conditions. As it can be seen from Table 1, the degree of DS desorption (ratio of diazonium salt quantity, found in the effluents after addition of extra NaNO2 and HCl and 15 min equilibration according to [22] and quantity of m-APAA in initial modified silica) increased significantly with the diazotation time and especially with HCl concentration. That is why in all further experiments, the diazotation was performed in optimized conditions, during 1 min, by 0.25 mol L−1 HCl, which resulted in only 5 % of reactant desorption. Importantly, no diazonium salt was detected in a rinsing buffer solution with pH 8; hence, the weakly alkaline media inhibits the DS desorption. Value of the concentration of active diazonium groups in SiO2–DS can be estimated from the value of its adsorption capacity to phenolic compounds, as it is discussed below.
Interaction of SiO2–DS with Phenolic Compounds
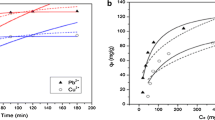
Common phenolic compounds are active in azo coupling reaction in the pH range 5–10. As the silica gel matrix becomes unstable at рН > 9, and the DS desorption takes place in the acidic media, the adsorption of phenolic compounds on SiO2 –DS was studied at pH 8.0. Dynamic adsorption isotherms (Fig. 2) were obtained in the conditions, which are typical for the SPE applications.
Initial light-yellow color of the SiO2 –DS changes to purple after interaction with phenolic compounds, confirming the formation of immobilized azo dye according to Scheme 3. At low loadings of phenolic compounds, nearly linear increase of adsorption with their concentration in effluent is observed, whereas at higher loadings, the isotherms come to saturation, corresponding to 60 μmol g−1 (Table 2). Probably, this value corresponds to the concentration of active diazonium groups on SiO2 –DS. At the same time, no coloration of the effluent was observed, so no desorption of the DS as well as the azo dyes takes place under dynamic adsorption experiments.
Surprisingly, the phenol did not adsorb on SiO2 –DS in noticeable amount under the selected conditions (pH 8); total adsorption capacity did not exceed 2 μmol g−1 and no significant coloration of SiO2 –DS was observed. Such a specificity of SiO2 –DS to polyhydroxy (resorcinol) and polyaromatic (naphthols) phenols can be explained by their higher activity in the azo coupling reaction comparing to the phenol. This last one can be adsorbed by SiO2 –DS but in different conditions: involving higher pH and wider contact time span.
High values of adsorption capacities and dynamic distribution coefficients (in a range of 102 g L−1, see Table 2) of SiO2 –DS to 1-naphtol, 2-naphtol, and resorcinol enable its application for pre-concentration of those pollutants from environmental water samples. For example, MAC of 1-naphthol in drinking water was reported as 100 μg L−1 [30]. SiO2 –DS packed (0.1 g) in SPE cartridge can be used for pre-concentration of 1-naphthol from approximately 250 mL of water solution having <5 MAC of 1-naphnol. The value of MAC for 1-naphthol in drinking water is 100 μg L−1. It means that 5 MAC values are equal to 500 μg L−1. Therefore, 250 mL of such a solution contains 125 μg of 1-naphthol. Adsorption capacity of SiO2 –DS is 6700 μg g−1, so in case of SPE cartridge packed with 0.1 g of the SiO2 –DS, the capacity is 670 μg. The ratio of the SPE capacity to the quantity of 1-naphtol in 250 mL is 670 μg:125 μg = 5,36. Therefore, under these conditions, the actual amount of the pollutant in a solution will be five times less than the adsorption capacity (6700 μg g−1).
Diffuse Reflectance UV Spectra of Immobilized Azo Compounds and 1-Naphthol Detection
High affinity of the SiO2 –DS to active phenols and bright color of the reaction products make this adsorbent prospective to develop solid-phase analytical reagent (SPAR) for DR-UV spectrophotometric determination of these substances. Figure 3 demonstrates the diffuse reflectance UV–vis spectra of SiO2 –DS after treatment with different phenolic compounds. As it was expected from the dynamic adsorption data, bright coloration of the adsorbent and corresponding intense bands in the visible spectrum range were observed only for active phenolic compounds: 1-naphthol (λ max = 530 nm), 2-naphthol (λ max = 490 nm), and resorcinol (λ max = 440 nm), whereas for less active phenol and catechol, the adsorbent coloration was moderate. Partial overlapping of the spectral bands of different azo dyes should result in significant overestimation of 1-naphtol concentration, determined by developed SPAR in the presence of 2-naphtol, resorcinol, or other azo coupling active compounds, whereas the influence of less active phenol and catechol should be much lower.
To find the optimal time of interaction between SiO2–DS and phenolic compounds, time dependencies of the analytical signal (i.e., the value of Kubelka-Munk function at λ max) were studied (Fig. 4). For the reaction times ≥2, the intensity of signal appeared nearly constant for all studied phenols, indicating high rates of their azo coupling according to Scheme 3. That is why 5 min equilibration time is sufficient for the formation of azo dye on the SiO2 surface, and this time interval was used in all further experiments.
As it can be seen from Figs. 3 and 4, the product of SiO2–DS interaction with 1-naphtol demonstrates the most intense color among all studied phenolic compounds, probably due to the highest extinction coefficient of the corresponding azo dye. Therefore, the highest sensitivity of 1-naphtol analysis procedure should be achieved. This factor together with the importance of 1-naphtol as an environmental pollutant allowed us to choose this compound as an analyte for further studies.
To study the effect of analyte dilution, samples of different volumes (25 mL–1 L) containing the same amount of 1-naphtol (0.5 μmol) were interacted with SiO2–DS (50 mg) in the conditions described above. Values of the analytical signal (F(R) at 530 nm) were compared. Variation of the solution volume in the 25–200-mL interval (500–4000 mL g−1 ratio of the solution volume to adsorbent mass, i.e., concentration factor) does not influence significantly on the signal; however, higher volumes result in its significant decrease (62 % from the initial value for 400 mL and 29 % for 1 L of solution).
Taking into account all the aforementioned data, 1-naphtol determination was performed under the following conditions of azo coupling: pH = 8, time of solid–liquid phase contact 5 min, and concentration factor equal to 2000 mL g−1. In the studied conditions, the calibration graph (i.e., F(R) concentration dependence) appeared perfectly linear in 0–0.8 mg L−1 1-naphtol concentration range (Fig. 5); the line equation is as follows:
LOD and limit of quantification (LOQ) values (20 and 66 μg L−1, correspondingly) were calculated from the parameters of calibration graph according to [31]. The LOD value corresponds to only 20 % of MAC for 1-naphthol in drinking water, reported in [30]; therefore, the sensitivity of the proposed method is sufficient for analysis of natural waters.
To evaluate the performance characteristics of the proposed method, model aqueous solutions of 1-naphtol were analyzed. Three parallel samples were tested for 100 and 600 μg L−1 solutions, and six parallel samples of as-prepared 300 μg L−1 solutions were analyzed twice with a 2-week period. The analysis results (confidence intervals, apparent recoveries, and RSDs) calculated according to [31] are presented in Table 3. These values demonstrate sufficient trueness, repeatability, and precision of the method and also the stability of prepared SPAR (SiO2–APAA) for at least 2 weeks storage.
Results of 1-naphthol analysis in the water from Lake Vyrlytsa (Kyiv), presented in Table 3, demonstrate an applicability of the proposed method for environmental water samples. The concentration of 1-naphtol in the lake water appeared below the sensitivity limits for both the proposed method and reference method described in [22]. Both tested methods give true (within the confidence interval) values of 1-naphtol concentration in the lake water spiked with MAC (100 μg L−1) of 1-naphtol.
To check the applicability of the proposed method for the analysis of 1-naphtol in different matrixes, the selectivity was investigated for following interferents: (i) inorganic salts and (ii) azo coupling active organic compounds. No influence of 100 mg L−1 of calcium, magnesium, sulfate, and carbonate ions, commonly present in environmental waters, was found. The results of 1-naphtol determination in the presence of organic interferents are shown in Table 4. Even high concentrations of phenol, resorcinol, and catechol did not interfere with 1-naphthol determination, probably due to their low activity in azo coupling with SiO2–DS, as discussed above. 2-Naphtol had no significant influence if present at the same concentration level as 1-naphtol analyte; however, its higher concentration resulted in a rise of the band at 490 nm in the DR-UV spectra and overestimated 1-naphtol analytical value as a result. Aromatic amines (except low-active 4-nitroaniline) also have significant interference due to their high activity in azo coupling.
Conclusions
The m-aminophenylarsonic acid could be efficiently immobilized on the silica gel surface via the ion exchange with covalently grafted propyl-trimethylammonium bromide groups. The diazotation of SiO2–APAA in slightly acidic conditions gives immobilized diazonium salt groups stable in neutral and slightly basic media. Resulted diazonium-silica can rapidly and selectively chemisorb the azo coupling active compounds, particularly phenols, forming bright-colored azo dyes. By measuring the color intensity of resulted azo dye, concentration of 1-naphtol in aqueous solutions can be detected. The sensitivity of such solid-phase spectrophotometric procedure is sufficient for the analysis of 1-naphtol pollutant in natural waters at the level above 0.2 MAC.
Abbreviations
- DR-UV:
-
UV diffuse reflection
- DS:
-
diazonium salt
- LOD:
-
limit of detection
- LOQ:
-
limit of quantification
- MAC:
-
maximum allowed concentration
- m-APAA:
-
m-aminophenylarsonic acid
- RSD:
-
relative standard deviation
- SiO2–DS:
-
silica modified with m-diazophenylarsonic acid
- SiO2–m-АPAA:
-
silica with covalently immobilized m-APAA groups
- SiO2–TMPA:
-
silica with covalently immobilized TMPA groups
- SPAR:
-
solid-phase analytical reagent
- TMPA:
-
trimethyl(3-trimethoxysilylpropyl)ammonium iodide
References
Zhu S, Zhang Q, Wang L, Chen J, Lian H (1999) Effect of ozonation of naphthalene derivatives on their elimination, TOC, and biodegradability. Bull Environ Contam Toxicol 63:101–108
Bolognesi C, Moretto A (2014) Genotoxic risk in rubber manufacturing industry: a systematic review. Toxicol Lett 230(Suppl 2):345–355
Olmo M, Laserna J, Romero D, Rohand J, Vilchez JL (1997) Determination of trace amounts of carbaryl in water by solid-phase laser-induced fluorescence. Talanta 44(Suppl 3):443–449
Rosenkranz HS, Klopman G (1990) Structural basis of mutagenecity of l-amino-2-naphthol-based azo dyes. Mutagen 5(Suppl 2):137–146
Chen CF, Hung IF, Chen RK (2006) Determination of 1-naphthol in urine by HPLC method polycyclic aromatic compounds. Polycyclic Aromat. Compd. doi: 10.1080/10406639908020612
Quintero MC, Silva M, Perez-Bendito D (1989) Simultaneous stopped-flow determination of 1- and 2-naphthol. Talanta 36(Suppl 7):717–722
Maggio RM, Damiani PC, Olivieri AC (2010) Four-way kinetic-excitation-emission fluorescence data processed by multi-way algorithms. Determination of carbaryl and 1-naphthol in water samples in the presence of fluorescent interferents. Anal Chim Acta 677(Suppl 2):97–107
Ortega-Algar S, Ramos-Martos N, Molina-Díaz A (2002) A continuous flow system combined with a sensing fluorimetric transductor for the determination of β-naphthol. Microchem J 73(Suppl 3):279–285
Jia G, Li L, Qiu J, Wang X, Zhu W, Sun Y, Zhou Z (2007) Determination of carbaryl and its metabolite 1-naphthol in water samples by fluorescence spectrophotometer after anionic surfactant micelle-mediated extraction with sodium dodecylsulfate. Spectrochim Acta A 67(Suppl 2):460–464
Hernandez-Lopez M, Algarra-Gonzalez M, Lopez-Molina MI (1999) Synchronous-derivative phosphorimetric determination of 1- and 2-naphthol in irrigation water by employing β-cyclodextrin. Talanta 49(Suppl 3):679–689
Penalva J, Puchades R, Maquieira A, Gee S, Hammock BD (2000) Development of immunosensors for the analysis of 1-naphthol in organic media. Biosens Bioelectron 15(Suppl 3–4):99–106
Velikorodnyi AA, Morosanova EI (2000) Silica-based xerogels modified with cobalt(III): determination of naphthols in solutions by solid-phase spectrophotometry. J Anal Chem 55(Suppl 8):724–731
Dmitrienko SG, Myshak EN, Runov VK, Zolotov YA (1995) Photometric determination of phenols with polyurethane foams. Chem Anal (Warsaw) 40(Suppl 1):291–298
Zhu PL, Liu CL, Liu MC (2003) Solid-phase microextraction from small volumes of sample in a glass capillary. J Chromatogr A 988(Suppl 1):25–32
Zolotov YA, Ivanov VM, Amelin VG (2002) Chemical test methods of analysis, book 36. In: Barceló D (ed) Comprehensive analytical chemistry. Elsevier science, Amsterdam, pp 1–336
Wang X, Chen C, Li J, Wang X (2015) Ozone degradation of 1-naphthol on multiwalled carbon nanotubes/iron oxides and recycling of the adsorbent. Chem Eng J 262:1303–1310
Evgen'eva MI, Garmonov SY, Evgen’eva II (2002) Test films for the determination of aromatic amines and hydrazines in aqueous solutions. J Anal Chem 57(Suppl 2):159–163
Tomilov YV, Kostyuchenko IV, Shulishov EV, Nefedov OM (2002) Formation of cyclopropylazoarenes in the azo coupling reactions of the cyclopropanediazonium ion with active aromatic compounds. Mendel Commun 12(Suppl 3):104–105
Fiamegos YC, Stalikas CD, Pilidis GA, Dados AE, Karayannis MI (2000) Synthesis and analytical applications of 4-aminopyrazolone derivatives as chromogenic agents for the spectrophotometric determination of phenols. Anal Chim Acta 403:315–323
Rouessac F, Rouessac A (2000) Chemical analysis: modern instrumental methods and techniques. Wiley, London
Jacobs WA, Heidelberger M, Rolf IP (1918) On nitro- and aminoaryl arsonic acids. J Am Chem Soc 40(Suppl 10):1580–1590
Murthy PK, Sulochana M, Naidu NV (2012) A novel spectrophotometric method for the determination of carbaryl in environmental samples. Der Pharmacia Sinica 3(Suppl 2):224–228
Katsaounos CZ, Paleologos EK, Giokas DL, Karayannis MI (2003) The 4-aminoantipyrine method revisited: determination of trace phenols by micellar assisted preconcentration. Int J Environ Anal Chem 83:507–514
Griffete N, Ahmad R, Benmehdi H, Lamouri A, Decorse F, Mangeney C (2013) Elaboration of hybrid silica particles using a diazonium salt chemistry approach. Colloids Surf A Physicochem Eng Asp 439:145–150
Bagheri H, Bayat P, Piri-Moghadam H (2013) Grafting the sol–gel based sorbents by diazonium salts: a novel approach toward unbreakable capillary microextraction. J Chromatogr A 1318:58–64
Zaitsev VN, Khalaf VA, Zaitseva GN (2008) Organosilica composite for preconcentration of phenolic compounds from aqueous solutions. Anal Bioanal Chem 391(Suppl 4):1335–1342
Alekseev SA, Zaitsev VN, Fraissard J (2003) Synthesis and structure of grafted layer of silicas modified with alkanesulfonic acid. Russ Chem Bull 52:364–369
Perrin DD, Dempsey B (1974) Buffers for pH and metal ion control. Chapman and Hall, London
Scott RPW (1993) Silica gel and bonded phases: their production, properties and use in LC. Wiley, New York
Murthy NBK, Raghu KJ (1988) Soil bound residues of carbaryl and 1-naphthol: release and mineralization in soil, and uptake by plants. Environ Sci Health B 23:575–585
Magnusson B, Örnemark U (2014) Eurachem guide: the fitness for purpose of analytical methods—a laboratory guide to method validation and related topics, 2nd edn.. ISBN 978-91-87461-59-0
Acknowledgements
We are thankful to the organizing committee of the Conference of Nanotechnology and Nanomaterials (NANO-2015), which was held in the framework of the FP7 project Nanotvinning, for the opportunity to make the presentation and publish our research paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing Interests
The authors declare that they have no competing interests.
Authors’ Contributions
NZ synthesized the silica modified with diazonium salt and carried out analytical procedures of research; SA synthesized the silica modified with quaternary ammonium; VZ has made substantial contributions to the analysis and interpretation of data; VR conceived the study and participated in its design and coordination. All authors read and approved the final manuscript.
Authors’ Information
NZ is a research scientist at the Analytical Chemistry Department, National Taras Shevchenko University, Kiev, Ukraine. Her primary interest is focused on adsorption mechanisms and analytical application of amorphous and highly ordered porous silica, modified by covalent coupling with organic functions. She received her master’s degree in Analytical Chemistry at National Taras Shevchenko University in 2005. She then carried out her graduate work with Prof. V. Zaitsev and A. Walcarius, at both the National Taras Shevchenko University and University of Lorraine, Nancy, France, specializing in the synthesis and characterization of sol–gel hybrid materials for analytical applications and earned her PhD degree in 2013.
SA is an associate professor of Analytical Chemistry Department at the National Taras Shevchenko University of Kyiv, Ukraine. He is a lecturer in Quantumn Chemistry, Christal Chemistry, and Chemistry of Complex compounds. He received his master’s degree in Inorganic Chemistry in 1998 from the National Taras Shevchenko University of Kyiv, Ukraine, and his PhD in Inorganic Chemistry from the same University in 2002. His current research interests are modification inorganic solids (silica, silicon carbide, nano-diamonds, and nano-ferrum particles) by covalent coupling with organic functional groups, methods of characterization of modified surface, and heterogeneous catalysis.
VZ is a professor of the National Taras Shevchenko University, Kiev, Ukraine. He is a founder and an editor-in-chief of the international journal Methods and Objects of Chemical Analysis. He has graduated from the National Taras Shevchenko University in 1981 as an inorganic chemist. In 1985, he received his PhD and in 1997, his Sc.D. degree (both from the National Taras Shevchenko University). Since 1984, he has been working in National Taras Shevchenko University as a young scientists, assistant, and associate Professor. In 2001, he has been habilitated to position of full professor. In 2009, he has been elected to the National Academy of Sciences of Ukraine as a correspondent member in the field of surface chemistry. He is a leading specialist in the field of surface chemistry of organo-mineral composite materials with aspects of their application in analytical chemistry, catalysis, and biomedicine. His research interests are preparation of new organo-mineral hybrid materials, studying their chemical properties, investigation of specific features of their chemical behavior, generalization of the results with the purpose of development of new tools and methods for application of such materials.
VR is an associate professor of the Analytical Chemistry Department at the National Taras Shevchenko University of Kyiv, Ukraine. She received her master’s degree in analytical chemistry in 2002 from National Taras Shevchenko University of Kyiv, Ukraine. She received her PhD in Analytical Chemistry from the same university in 2006. The areas of her scientific interests are the sample preparation in chromatography, high-performance liquid chromatography, LC-MS, and GC-MS. Under the supervision of Prof. Vladimir Zaitsev, Dr. V. Raks designed and implemented an Analytical Skills Training Course for the Organization for the Prohibition of Chemical Weapons. Dr. V. Raks had industry training in the Open Joint-Stock Company “Rivneazot” in Rivne, Ukraine, 2012; training at BASF in Ludwigshafen, Germany, 2011; pilot plant training at the University of Surrey, the United Kingdom, 2011.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zaitseva, N., Alekseev, S., Zaitsev, V. et al. Solid-Phase Spectrophotometric Analysis of 1-Naphthol Using Silica Functionalized with m-Diazophenylarsonic Acid. Nanoscale Res Lett 11, 149 (2016). https://doi.org/10.1186/s11671-016-1356-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s11671-016-1356-2