Abstract
Cu2+1O coated Si nanoparticles were prepared by simple hydrolysis and were investigated as an anode material for lithium-ion battery. The coating of Cu2+1O on the surface of Si particles remarkably improves the cycle performance of the battery than that made by the pristine Si. The battery exhibits an initial reversible capacity of 3063 mAh/g and an initial coulombic efficiency (CE) of 82.9 %. With a current density of 300 mA/g, its reversible capacity can remains 1060 mAh/g after 350 cycles, corresponding to a CE ≥ 99.8 %. It is believed that the Cu2+1O coating enhances the electrical conductivity, and the elasticity of Cu2+1O further helps buffer the volume changes during lithiation/delithiation processes. Experiment results indicate that the electrode maintained a highly integrated structure after 100 cycles and it is in favour of the formation of stable solid electrolyte interface (SEI) on the Si surface to keep the extremely high CE during long charge and discharge cycles.
Similar content being viewed by others
Background
The application of lithium-ion battery is playing an important role in the development of portable electric and electro vehicles. Graphite has dominated as the anode material of lithium-ion batteries and has been commercialized for many years due to its excellent behaviour during prolonged charge/discharge cycles. However, the theoretical capacity of graphite is limited to 372 mAh/g, which is low relative to the requirement of high energy density application fields [1]. To develop a low-cost electrode material with a high energy capacity is of great significance to improve the performance of products that use rechargeable batteries. Crystal Si has attracted much attention as a possible anode candidate due to the much higher lithium storage capacity (4200 mAh/g, about ten times higher than graphite), low lithium alloying/dealloying potential, long discharge plateau and natural abundance [2]. However, Si-based anodes also face grand challenges due to the large volume expansion (about 400 %) of the Si particles during lithiation/delithiation processes. It results in pulverization, breaks the electrical contact of the electrode structure and brings in great capacity decay [3]. The lack of electrical contacts between Si particles or between Si and current collector even makes capacity fading worse. Many investigations have been done to accommodate this severe volume expansion, mainly including novel nanostructured Si such as Si wires [4], Si tubes [5], porous Si thin films [6] and nest-like Si nanospheres [7] or multiphase composites consisting of active Si and other active/inactive phases [8, 9]. Among these materials, Si-based composites containing Si and coating other ductile materials as buffer were conducted to reduce volume expansion. Carbon, metal, metal oxide and conducting polymers are used as the shell materials, which can act as both conducting and mechanical supporting material [10–14]. Among them, the coating of metal and metal oxide on Si electrodes will be a good way to improve their electrochemical performances. Due to the introduction of the surplus metal Cu in the coating layer, it has been proved to greatly increase the electrical conductivity in a Si-based anode system and help buffer the volume changes during insertion/extraction processes of lithium ions [15–17].
In this study, a scalable, chemical approach for synthesizing Cu2+1O-coated polycrystalline Si particles through a hydrolysis method is reported. The Cu2+1O-coated particles were utilized as anode material for lithium-ion battery. The coating of Cu2+1O on Si particles reduced charge transfer resistance, increased the reversible capacity and improved the tolerance for volume changes during lithiation/delithiation processes.
Our investigation revealed that Cu2+1O-coated Si electrode showed significantly improved cycle performance even after 350 cycles. It maintained an extremely integrated structure after 100 cycles. It is confirmed that the Cu2+1O coating on Si enhanced the conductivity and buffered the volume changes during insertion/extraction processes of lithium ions, leading to a highly stable cycle performance.
Methods
Experimental procedures
Si particles of 80 nm were treated by hydrofluoric acid (HF, 10 %) for 10 min and then centrifuged and washed by deionized water three times. Ammonium formate (6.3 g) was dissolved in deionized water (500 mL), and formic acid (0.5 mL) was added. After copper sulphate (0.007 mol/L) was added and dissolved, the Si particles treated by HF were added and the reaction was maintained at a constant temperature of 70 °C for 2 h. After that, the material was centrifuged and washed by deionized water three times and heated in vacuum drying oven overnight. Then, the composite particles, super P, graphite, VGCF and polyacrylic acid were mixed for 6:1:1:1:1 and stirred for 6 h to be evenly mixed. The slurry was coated on a copper foil with a thickness of 100 μm, followed by heat treatment at 110 °C in a vacuum drying oven overnight, and then, the anode was ready for cell assembling.
Characterization
The morphologies of the pristine Si particles and Cu2+1O-coated Si particles were investigated by transmission electron microscope (TEM), which was performed with a small amount of sample dispersed in ethanol and dropped into Au TEM grid for analysis. The structures were characterized by X-ray diffraction (XRD) to find the crystallinity and composition of the sample, and the radial data (2θ) were integrated over 10°–70°.
Electrochemical measurements
Coin-type half cells (2025R type) with lithium foils as counter electrodes were assembled in a glove box (Mikrouna Super 1220/750) under an argon atmosphere. The electrolyte used was LiPF6 (1 M) in ethylene carbonate-methyl ethyl carbonate (30:70 vol%), with vinylene carbonate and fluoroethylene carbonate as additives. Glass fibre was employed to stabilize the coin system. The coin-type half cells were galvanostatic cycled (with a constant current of 300 mAh/g and cut-off voltages of 0.01 and 1.2 V at 25 °C) on land battery test system. The cyclic voltammetry (CV) curves (in the range of 10 mV to 2.6 V and at a rate of 0.2 mV/s) and electrochemical impedance spectroscopy (EIS, in the range of 100,000 to 0.01 Hz at a magnitude of 0.05 mV) were measured with an electrochemical workstation (PGSTAT302N, Autolab).
Results and discussion
Figure 1 shows the TEM images of the samples. Clearly, the pristine Si particles have a smooth surface with the size of about 80 nm as shown in Fig. 1a. Their surface becomes rough after HF treatment due to the reaction of HF with SiO2 on the surface of Fig. 1b. This surface roughness is proved to be beneficial for the adherence of Cu2+1O with Si during hydrolysis. It can be found that the nano-sized irregular-shaped Cu2+1O particles decorate on the Si sample surface after hydrolysis (Fig 1c). The energy dispersive X-Ray spectroscopy (EDS) examination confirms the existence of Si, Cu, O, Au and C in Cu2+1O-coated Si particles. Here, Au and C contents are from the TEM grid, Cu is from the deposition of Cu2+1O on the surface of HF treated Si particles and O is from both Cu2+1O and the remaining SiO2. The weight ratio of Cu2+1O and Si in the final composite is about 1:3.
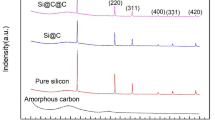
XRD results confirm that the successful coating of Cu2+1O on the surface of Si particles by the chemical precipitation during the hydrolysis and drying process since all peaks marked in Fig. 2 match well with Bragg peaks of Si and Cu2+1O, and the peaks of Si are consistent with the peaks of pristine Si XRD results.
These materials were then used as the anode and assembled into a lithium-ion battery. Experimental results reveal that the coating of Cu2+1O on Si particles remarkably improved the cycle performance of the battery. It has an initial reversible capacity up to 3063 mAh/g and an initial coulombic efficiency (CE) of 82.9 % (Fig. 3a). With a current density of 300 mA/g, the reversible capacity of the composite electrode remains 62.8 % after 100 cycles, as 1923.5 mAh/g with a CE ≥99.7 %, and even after 350 cycles, the reversible capacity is more than 1060 mAh/g with a CE ≥99.8 %. As comparison, the reversible capacity of the composite electrode with 80-nm pristine Si which is purchased without any treatment decreases significantly, which remains only 19.2 % after 100 cycles, as 613.6 mAh/g with the same current density of 300 mA/g (Fig. 3a). What is more, the cycle performance of Cu2+1O-coated Si electrode with a higher current density of 800 mA/g is conducted. It has an initial reversible capacity up to 3329 mAh/g and an initial CE of 83.5 % (Fig. 3b). Even after 100 cycles, the reversible capacity was more than 1110 mAh/g with a CE ≥97 %. We have made the parallel tests for each cell, and the repeatability and consistency of the cell performance is good. These results reveal that the coating of Cu2+1O on Si as electrode has greatly improved cycle performance than pristine Si electrode. Compared to the pristine Si electrode, the high capacity retention and high CE of Cu2+1O coated Si electrode is because the coating of Cu2+1O enhances the conductivity and effectively suppresses the volume changes during insertion/extraction processes of lithium ions and helps the formation of stable solid electrolyte interface (SEI) [18, 19] .
In order to investigate the rate performance of Cu2+1O-coated Si electrode, galvanostatic measurements were conducted from 0.1 C (1 C = 1000 mA/g) to 2 C (Fig. 4) with current density of 0.1, 0.3, 0.5, 1, 2 and 0.1 C (each density of current for ten cycles). Obviously, the Cu2+1O-coated Si electrode has better rate performance with capacities of 2984.7, 2293.3, 2056.4, 1579.5 and 931.9 mAh/g at the first cycle of each current density. It rises again up to 2310.9 mAh/g at low current density (0.1 C). This may be related to the activation of the unreacted inner region of silicon, which is generally accepted for Si anodes operated under high rate conditions [20]. As comparison, pristine Si electrode has lower capacities of 2966.5, 2135.2, 1887.5, 1282.8 and 637.8 mAh/g, at the first cycle of each current density, and it rises back up to 2170.3 mAh/g at 0.1 C.
Figure 5 shows the capacity of Cu2+1O-coated Si electrode as the function of the voltage. The charge and discharge profiles of Cu2+1O-coated Si electrode for the first, second, third, fifth, tenth, 20th and 50th cycles are tested with a current density of 300 mA/g. The wide plateau at about 0.2 V in the discharge process is original from the formation of LixSi alloy [21, 22]. The initial discharge and charge capacities are 3694.8 and 3063.1 mAh/g, respectively, with a CE of 82.9 %. During the second cycle, a large reversible capacity of 2979.9 mAh/g is obtained, with a CE of 95.3 %. Even during the 50th cycle, the reversible capacity still remains 2227.5 mAh/g, with a high CE of 99 %. The stable cycle performance is benefiting from the Cu2+1O coating layer, which contributes to the increased conductivity and suppression of volume changes of Si during insertion/extraction processes of lithium ions.
In order to further elucidate the effect of Cu2+1O as conductive material, the impedance spectroscopies were investigated for the pristine Si and Cu2+1O-coated Si electrodes (as prepared and after 100 charge/discharge cycles), and the results are showed in Fig. 6. All of the electrodes used in the impedance test have the same thickness and area, which are 100 μm and π*(1.5/2)2 cm2, respectively, so only resistance is used for comparison. Apparently, the diameter of the semicircle for the Cu2+1O-coated Si electrode is smaller than that of the pristine Si before cycling, indicating the smaller surface resistance and charge transfer resistance as shown in Fig. 6a. Thereafter, the impedance test after 100 cycles indicates that pristine Si shows a larger charge transfer resistance as shown in Fig. 6b. The possible reason is that lithium ion conduction in the SEI layers and charge transfer at the electrode/electrolyte interface are hindered by the increased defects due to the volume changes and structure failure during continued cycling. In contrast, the semicircle, corresponding to the Cu2+1O-coated Si, shows increased but lower charge transfer resistance after 100 cycles as also shown in Fig. 6b. This indicates the Cu2+1O deposited on the Si surface is beneficial for the formation of low resistance SEI layer [23].
The surface and the cross section SEM images of Cu2+1O-coated Si electrode and pristine Si electrode after 100 charge and discharge cycles are showed in Fig. 7. The surface of Cu2+1O-coated Si electrode is smooth and integrated with only some unconspicuous cracks after 100 cycles (Fig. 7a). And the cross section of Cu2+1O-coated Si electrode is keeping entirely integrated (Fig. 7c). For comparison, pristine Si electrode becomes extremely rough and has obvious cracks both in the surface (Fig. 7b) and the cross section images (Fig. 7d). We believe that the Cu2+1O coating layer suppress the volume changes of Si during insertion/extraction processes of lithium ions. It is helpful for the electrode keeping integrated and electrical contact during long cycles [24, 25].
Conclusions
The coating of Cu2+1O on Si particles prepared by simple hydrolysis reaction remarkably improved the cycle performance of the battery with an initial reversible capacity of 3063 mAh/g and a CE of 82.9 %. The reversible capacity of the composite remained as 1923.5 mAh/g, 62.8 % of its initial capacity after 100 cycles and 1060 mAh/g after 350 cycles, corresponding to a CE of ≥99.8 % with a current density of 300 mA/g. The composite is easy for mass production. The corresponding electrode maintained extremely good structure and had excellent electrochemical performance during cycling. This is attributed to the enhanced conductivity and elasticity benefited from the Cu2+1O coating layer.
Abbreviations
- CE:
-
coulombic efficiency
- CV:
-
cyclic voltammetry
- EIS:
-
electrochemical impedance spectroscopy
- HF:
-
hydrofluoric acid
- SEI:
-
solid electrolyte interface
- TEM:
-
transmission electron microscope
- XRD:
-
X-ray diffraction
References
Mohri M, Yanagisawa N, Tajima Y, Tanaka H, Mitate T, Nakajima S, Yoshida M, Yoshimoto Y, Suzuki T, Wada H (1989) Rechargeable lithium battery based on pyrolytic carbon as a negative electrode. J Power Sources 26:545–551
Kasavajjula U, Wang CS, Appleby A (2007) Nano- and bulk-silicon-based insertion anodes for lithium-ion secondary cells. J Power Sources 163:1003–1039
Timmons A, Dahn JR (2007) Isotropic volume expansion of particles of amorphous metallic alloys in composite negative electrodes for Li-ion batteries. J Electrochem Soc 154:A444–A448
Föll H, Hartz H, Ossei-Wusu E, Carstensen J, Riemenschneider O (2010) Si nanowire arrays as anodes in Li ion batteries. Physica Status Solid-Rapid Research Letters 4:4–6
Song T, Xia JL, Lee J, Lee JH, Lee DH, Kwon MS, Choi JM, Wu J, Doo SK, Chang H, Park WI, Zang DS, Kim H, Huang YG, Hwang KC, Rogers J, Paik U (2010) Arrays of sealed silicon nanotubes as anodes for lithium-ion batteries. Nano Lett 10:1710–1716
Yu CJ, Li X, Ma T, Rong JP, Zhang RJ, Shaffer J, An YH, Liu Q, Wei BQ, Jiang HQ (2012) Silicon thin films as anodes for high-performance lithium-ion batteries with effective stress relaxation. Adv Energy Mater 2:68–73
Ma H, Cheng F, Chen JY, Zhao JZ, Li CS, Tao ZL, Liang J (2007) Nest-like silicon nanospheres for high capacity lithium storage. Adv Mater 19:4067–4070
Kim SO, Manthiram A (2015) A facile, low-cost synthesis of high-performance silicon-based composite anodes with high tap density for lithium-ion batteries. J Mater Chem A 3:2399–2406
He DF, Bai FJ, Li L, Shen LX, Shen LM, Kung HH, Bao NZ (2015) Fabrication of sandwich structured Si nanoparticles graphene nanocomposites for high-performance lithium-ion batteries. Electrochim Acta 169:409–415
Murugesan S, Harri JT, Korgel BA, Stevenson KJ (2012) Copper-coated amorphous silicon particles as an anode material for lithium-ion batteries. Chem Mater 24:1306–1315
Cetinkaya T, Uysal M, Guler MO, Akbulut H, Alp A (2014) Improvement cycle ability of core-shell silicon copper composite electrodes for Li-ion batteries by using electroless deposition of copper on silicon powders. Powder Technol 253:63–69
Tang H, Zhang J, Zhang YJ, Xiong QQ, Tong YY, Li Y, Wang XL, Gu CD, Tu JP (2015) Porous reduced graphene oxide sheet wrapped silicon composite fabricated by steam etching for lithium-ion battery application. J Power Sources 286:431–437
Sun LM, Wang XH, Susantyoko RA, Zhang Q (2014) Copper-silicon core-shell nanotube arrays for free-standing lithium-ion battery anodes. J Mater Chem A 2:15294–15297
Kim GP, Nam I, Park S, Park J, Yi J (2013) Preparation via an electrochemical method of graphene films coated on both sides with NiO nanoparticles for use as high-performance lithium ion anodes. Nanotechnology 24:475402
Li CL, Zhang P, Jiang ZY (2015) Effect of nano Cu coating on porous Si prepared by acid etching Al-Si alloy powder. Electrochim Acta 161:408–412
Oh SM, Jae WK, Ji HR, Lee KT (2005) Improvement of silicon powder negative electrodes by copper electroless deposition for lithium secondary batteries. J Power Sources 147:227–233
Morales J, Sanchez L, Bijani S, Martnez L, Gabs M, Ramos-Barrado JR (2005) Electrodeposition of Cu2O: an excellent method for obtaining films of controlled morphology and good performance in Li-ion batteries. Electrochem Solid-State Lett 8:A159–A162
Ling M, Zhao H, Xiao XC, Shi FF, Wu MY, Qiu JX, Li S, Song XY, Liu G, Zhang SQ (2015) Low cost and environmentally benign crack-blocking structures for long life and high power Si electrodes in lithium ion batteries. J Mater Chem A 3:2036–2042
Tao HC, Yang XL, Zhang LL, Ni SB (2015) Reduced graphene oxide/porous Si composite as anode for high-performance lithium ion batteries. Ionics 21:617–622
Wang N, Hang T, Ling HQ, Hu AM, Li M (2015) High-performance Si-based 3D Cu nanostructured electrode assembly for rechargeable lithium batteries. J Mater Chem A 3:11912–11919
Xu YH, Yin GP, Ma YL, Zuo PJ, Cheng XQ (2010) Nanosized core/shell silicon@carbon anode material for lithium ion batteries with polyvinylidene fluoride as carbon source. J Mater Chem 20:3216–3220
Huang R, Fan X, Shen W, Zhu J (2009) Carbon-coated silicon nanowire array films for high-performance lithium-ion battery anodes. Appl Phys Lett 95:133119
Huang XK, Mao SM, Chang JB, Hallac PB, Fell CR, Luo YT, Metz B, Jiang JW, Chen JH (2015) Improving cyclic performance of Si anode for lithium-ion batteries by forming an intermetallic skin. RSC Adv 5:38660–38664
Polat BD, Keles O (2015) The effect of copper coating on nanocolumnar silicon anodes for lithium ion batteries. Thin Solid Films 589:543–550
Wang XL, Li G, Hassan FM, Li M, Feng K, Xiao XC, Chen ZW (2015) Building sponge-like robust architectures of CNT-graphene-Si composites with enhanced rate and cycling performance for lithium-ion batteries. J Mater Chem A 3:3962–3967
Acknowledgements
This work was supported in part by the National Thousand Talents Program of China, the bilateral collaboration project between Chinese Academy of Sciences and Japan Society for the Promotion of Science (Grant no. GJHZ1316), Beijing Natural Science Foundation (Grant no. 2142031), Beijing Municipal Science and Technology Commission project (Grant no. Z141100003814002), the Major State Basic Research Development Program of China (Grant no. 2013CB632103), the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry, Key Laboratory of Nanodevices and Applications, Suzhou Institute of Nano-Tech and Nano-Bionics, Chinese Academy of Sciences and China Postdoctoral Science Foundation funded project (Grant no. 2015M581150).
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
JZ and CZ carried out the experiments, performed the statistical analysis and drafted the manuscript. SW, ZL, JZ, YZ, CX and BC participated in parts of experiments and the analysis of the results. CL is the corresponding author, who conceived the study and participated in the design of the study, data analysis and the modification of the manuscript. CL and BC agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors read and approved the final manuscript.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhang, J., Zhang, C., Wu, S. et al. Cu2+1O coated polycrystalline Si nanoparticles as anode for lithium-ion battery. Nanoscale Res Lett 11, 214 (2016). https://doi.org/10.1186/s11671-016-1426-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s11671-016-1426-5