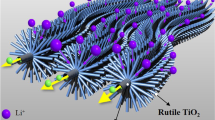
Abstract
In this paper, rutile TiO2 nanorod arrays are fabricated by a template-free method and proposed as a promising anode for aqueous Li-ion battery. The as-prepared TiO2 nanorod arrays exhibited reversible Li-ion insertion/extraction ability in aqueous LiOH electrolyte. Moreover, galvanostatic charge/discharge test results demonstrated that the reversible capacity of TiO2 nanorods could reach about 39.7 mC cm−2, and 93.8 % of initial capacity was maintained after 600 cycles at a current density of 1 mA cm−2 (=240 C rate), indicating excellent cycling stability and rate capability.
Similar content being viewed by others
Background
The continuously increasing demand for electronic devices, particularly the large-scale energy storage system (ESS) and electric vehicles (EVs), has forced researchers to investigate new classes of materials for replacement of the conventional Li-ion batteries (LIBs) and Ni-MH batteries [1]. In the mid of 1990s, a new type of Li-ion battery with aqueous electrolyte proposed by L. Wu et al. has intrigued many researchers due to its inherent safety, low-cost, and similar system as LIBs [2]. Compared with non-aqueous LIBs, the safety problem of aqueous Li-ion batteries (ALIBs) is fundamentally resolved, the ion conductivity of the electrolyte is enhanced by several magnitudes, and the rigorous assembly conditions are avoided, so the cost is greatly reduced [3, 4]. Unfortunately, ALIBs exhibited a poor cycling performance because of the complicated Li insertion/extraction process in aqueous solution, which would aggravate the dissolution of active materials and deterioration of crystal structure [3, 4]. Moreover, the decomposition of water also accelerates the fading of capacity and limits the working voltage of ALIBs.
Recently, the working voltage of ALIBs has been obviously improved using “water-in-salt” electrolyte, which attracts more and more researcher focusing on ALIBs [5, 6]. To obtain high-performance ALIB, advanced electrode materials are indispensable. Many strategies have been attempted to improve the electrochemical properties [3, 4], such as surface coating with conductive polymer or high-quality carbon, preparation of porous active materials [7], and fabrication of nanostructured active materials (e.g., nanoflake, nanotube, nanowire, and so on.) [8–14] Among these strategies, fabricating nanostructured electrodes demonstrate unique advantages over others [11, 12]. It is generally believed that nanostructured materials play an important role in electrochemical performance because of the high-specific surface area and short diffusion path which could enhance the electrochemical behavior of the applied battery [8, 14, 15].
In recent years, titanium dioxide (TiO2) has attracted considerable academic and practical interests [10, 16]. Its semi-conducting properties together with the excellent chemical stability make it an excellent candidate for solar cells, electrochromic devices, and rechargeable batteries [17–19]. In particular, nanostructured TiO2 for non-aqueous LIB anode has been extensively studied [20]. Recently, Manickam et al. proved that LixTiO2 is electrochemically reversible in LiOH aqueous solution [21]. Reiman et al. demonstrated the reversibility of Li x TiO2 in aqueous solution with the aid of Dahn’s calculation of the HER potential vs. Li+/Li and fabricated TiO2 nano-film, which showed superior Li-ion storage properties [22]. Wu et al. prepared TiO2 nano-film via anodic electrodeposition method and obtained the diffusion coefficient through CVs method [14]. Note that the abovementioned TiO2 as anode for ALIBs are all concentrated on anatase phase. As we know, the phase plays an important role on the electrochemical properties [23]. Moreover, the one-dimensional nanosized materials (such as nanorod, nanotube, nanofiber, and so on [11, 12, 16, 24]) with faster kinetics and higher rate capability due to the large surface-to-volume ratio are attracting more and more attention [11].
In the present work, we first reported another phase of TiO2 (rutile phase) as anode for ALIBs. Rutile TiO2 nanorod array was deposited on fluorine-doped tin oxide (FTO) substrate via a template-free strategy, and the electrochemical performance of the as-synthesized TiO2 nanorod array in LiOH electrolyte was investigated in detail. It is interesting to note that the rutile TiO2 nanorod array exhibited excellent superior rate capability and excellent cycling stability, making it a promising anode candidate for ALIBs.
Methods
In a typical synthesis, as a similar process described in the literature [16], 10 mL of toluene was mixed with 1 mL of 1 M titanium tetrachloride in toluene (97 % Aldrich) and 0.1 mL of tetrabutyl titanate in a Teflon-lined stainless steel autoclave (50 mL). The mixture was stirred at ambient conditions for 1 min followed by the addition of 1 mL of hydrochloric acid (37 wt %). After stirring for another 5 min, one piece of FTO glass substrate (3 cm × 1 cm) was placed at an angle against the wall of the Teflon liner. The FTO glass was initially cleaned by sonication in acetone, subsequently immersed in a 1 M NaOH solution for 10 min, and finally rinsed with ethanol and deionized water. Solvothermal synthesis was conducted at 180 °C for 2 h in an electric oven. After that, the autoclave was cooled to room temperature for approximately 70 min. Then the TiO2/FTO substrate was washed thoroughly with ethanol and allowed to dry under room temperature.
The crystal structure of the as-prepared film was examined by X-ray diffraction (XRD). The XRD patterns were recorded in a Bruker-AXS Micro-diffractometer (D8 ADVANCE) with Cu Kα radiation (λ = 1.5406 Å) from 10° to 70° at a scanning speed of 0.33° min−1. X-ray tube voltage and current were set at 40 kV and 40 mA, respectively. Morphological information was attained from scanning electron microscopy (SEM; HITACHI S-4800).
For electrochemical studies, electrical contact was made to some exposed FTO at the edge of the TiO2-coated slide, and the area of the TiO2 is 1 cm2. Conventional cyclic voltammetry (CV) and galvanostatic charge/discharge of the electrode were characterized by using a three-electrode cell in which an Hg/HgO and Pt sheet electrode were used as reference electrode and counter electrode, respectively. De-oxygenated 1 M aqueous LiOH solution was used as electrolyte. All electrochemical studies were conducted using CHI440a electrochemical station at room temperature.
Results and Discussion
XRD pattern of the as-prepared sample is displayed in Fig. 1. As can be seen, the diffraction peaks, except for those with the asterisk which are due to the FTO substrate, could be well indexed to the tetragonal rutile TiO2 phase (JCPDS No. 21-1276) [16]. That is, rutile TiO2 could be deposited on the FTO substrate via a solvothermal process. The SEM images of top view and cross-sectional view of a typical TiO2 nanorod array sample are shown in Fig. 2. The images demonstrate that the surface of the FTO substrate is covered uniformly with aligned TiO2 nanorods. The top surface of the nanorods appears to be flat tetragonal crystallographic planes while the side surface is smooth. Nanorod arrays are nearly perpendicular to the FTO substrate. These results are consistent with the morphology of rutile nanorod arrays reported recently [16] and indicate that the TiO2 nanorod arrays have been successfully grown on FTO substrate. As determined from SEM images, the average diameter and length of nanorods are around 100 nm and 1.2 μm, respectively. It is well known that the application of 1D nanostructure in electrode design could provide high surface area and short ion path length, leading to higher capacities at high charge/discharge rates [11].
CV curves of TiO2 nanorod array between −0.40 and −1.60 V (vs. Hg/HgO) with different scanning rates (5, 10, 25, 50, and 100 mV/s) in 1 M LiOH solution are shown in Fig. 3. Clearly, CV curves demonstrate a good Li-ions insertion/extraction reversibility of TiO2 nanorod array in LiOH solution. With the increase of scanning rate, the potential separation between the cathodic peak and anodic peak of TiO2 nanorod array become more and more larger, suggesting the increased polarization. The CV results at various scanning show the linear relationship (inset of Fig. 3) between the peak current and the square root of scan rate in anodic processes. The good linearity of anodic peaks reveals that the Li-ions insertion into the sample is a semi-diffusion controlled process, which can be treated as a quasi-reversible system with good reversibility [12, 14, 25–27].
Galvanostatic charge/discharge curves of TiO2 nanorod array at different current densities in LiOH solution are displayed in Fig. 4. A charge/discharge plateau around −1.4 V vs. Hg/HgO is observed. Table 1 summarizes the specific capacity of TiO2 nanorod array at different current densities. As seen, the specific capacity gradually decreases with the increasing of charge/discharge current density. However, the coulombic efficiency shows a reverse change. As we know, the decomposition of H2O during the charge/discharge capacity is the main reason for the low coulombic efficiency of ALIB [3, 7]. Although the Li insertion potential of TiO2 is relatively low, it is possible to achieve the Li-ion insertion/extraction of TiO2 before the hydrogen evolution reaction (HER) by tuning the electrolyte systems and the current collectors. In this work, we used the LiOH solution, rather than neutral Li2SO4 or LiNO3 as electrolyte, in which the HER would be well suppressed. In addition, increasing the current density could further reduce the decomposition of water. At a low current density, the HER is serious, which results in a low coulombic efficiency (65.8 % for 1 mA cm−2). On increasing the current density, the polarization of HER increases and reaction time shortens, resulting in a much higher coulombic efficiency (95.2 % for 8 mA cm−2).
A constant current charge/discharge measurement of TiO2 nanorod array at 1 mA cm-2 was performed and some of charge/discharge curves are shown in Fig. 5. This result is significant when we realize that it is for a macroscopically planar electrode surface, so that much larger current densities can be expected for electrodes in which the same film thickness is distributed over a microscopically roughened current collector as in modern battery electrodes. Accordingly, since the actual TiO2 film thickness here was only 1.2 μm, we can predict that an external current density of nearly 1 A cm-2 should be possible for a typical battery electrode thickness of 120 μm.
Figure 6 displays the capacity vs. cycle number data for the TiO2 nanorod array electrode. These data were obtained from galvanostatic charge/discharge measurement over the potential range of −0.2~−1.6 V at a current density of 1 mA cm-2. The initial discharge capacity obtained over this potential window was 39.5 mC cm−2. After 600 cycles, the discharge capacity only decreased by 7 % of the original value, indicating the superior cycling stability of TiO2 nanorod array in LiOH solution. This represents another unique advantage of the nanostructured electrode.
Generally, low utilization of rutile TiO2 nanorod was demonstrated in aqueous solution in comparison with that observed in organic electrolyte due to the narrow electrochemical stability window of aqueous solution. Anyway, this research indicates that Li ions are partially electro-reversible in rutile TiO2, and the unique nanostructure facilitates the electrochemical behavior significantly.
Conclusions
Rutile TiO2 nanorod array growing on FTO substrate was obtained successfully. CV results showed that Li-ion intercalation into/extraction from rutile TiO2 electrode can be occurred in LiOH solution. The TiO2 nanorod array demonstrated a good rate capability up to 39.7 mC cm−2 at 1 mA cm−2, much higher than that in organic electrolyte. Moreover, excellent cycling stability with only 7 % capacity loss after 600 cycles was observed for TiO2 nanorod array electrode. This work implies that nanostructured TiO2 array could be used as a promising anode for advanced aqueous Li-ion battery.
Abbreviations
- ALIBs:
-
Aqueous Li-ion batteries
- ESS:
-
Energy storage system
- EVs:
-
Electric vehicles
- LIBs:
-
Li-ion batteries
- TiO2 :
-
Titanium dioxide
References
Armand M, Tarascon JM (2008) Building better batteries. Nature 451:652–657
Li W, Dahn JR, Wainwright DS (1994) Rechargeable lithium batteries with aqueous electrolytes. Science (New York, NY) 264:1115–1118
Tang W, Zhu Y, Hou Y et al (2013) Aqueous rechargeable lithium batteries as an energy storage system of superfast charging. Energy Environ Sci 6:2093–2104
Wang Y, Yi J, Xia Y (2012) Recent progress in aqueous lithium-ion batteries. Adv Energy Mater 2:830–840
Suo L, Borodin O, Gao T et al (2015) “Water-in-salt” electrolyte enables high-voltage aqueous lithium-ion chemistries. Science 350:938–943
Yamada Y, Usui K, Sodeyama K et al (2016) Hydrate-melt electrolytes for high-energy-density aqueous batteries. Nat Energy 1:16129
Luo JY, Xia YY (2007) Aqueous lithium-ion battery LiTi2(PO4)3/LiMn2O4 with high power and energy densities as well as superior cycling stability. Adv Funct Mater 17:3877–3884
Sun D, Jin G, Wang H et al (2014) Aqueous rechargeable lithium battery using NaV6O15 nanoflakes as a high performance anode. J Mater Chem A 2:12999–13005
Zhou D, Liu S, Wang H et al (2013) Na2V6O16·0.14H2O nanowires as a novel anode material for aqueous rechargeable lithium battery with superior cycling performance. J Power Sources 227:111–117
Zhao Z, Tian J, Sang Y et al (2015) Structure, synthesis, and applications of TiO2 nanobelts. Adv Mater 27:2557–2582
Wang X, Li Z, Shi J et al (2014) One-dimensional titanium dioxide nanomaterials: nanowires, nanorods, and nanobelts. Chem Rev 114:9346–9384
Lee K, Mazare A, Schmuki P (2014) One-dimensional titanium dioxide nanomaterials: nanotubes. Chem Rev 114:9385–9454
Tian J, Zhao Z, Kumar A et al (2014) Recent progress in design, synthesis, and applications of one-dimensional TiO2 nanostructured surface heterostructures: a review. Chem Soc Rev 43:6920–6937
Wu M-S, Wang M-J, Jow JJ et al (2008) Nanostructured silver-catalyzed nickel foam cathode for an aluminum-hydrogen peroxide fuel cell. J Power Sources 185:1420–1424
Sun D, Jiang Y, Wang H, Yao Y et al (2015) Advanced aqueous rechargeable lithium battery using nanoparticulate LiTi2(PO4)3/C as a superior anode. Sci Rep 5:10733
Liu B, Aydil ES (2009) Growth of oriented single-crystalline rutile TiO2 nanorods on transparent conducting substrates for dye-sensitized solar cells. J Am Chem Soc 13:3985–3990
Shaban YA, Khan SUM (2008) Visible light active carbon modified n-TiO2 for efficient hydrogen production by photoelectrochemical splitting of water. Int J Hydrogen Energy 33:1118–1126
Nowotny J, Bak T, Nowotny MK et al (2007) Titanium dioxide for solar-hydrogen I. Functional properties. Int J Hydrogen Energy 32:2609–2629
Kumari S, Chaudhary YS, Agnihotry SA et al (2007) A photoelectrochemical study of nanostructured Cd-doped titanium oxide. Int J Hydrogen Energy 32:1299–1302
Liu S, Ye S, Li C et al (2011) Rechargeable aqueous lithium-ion battery of TiO2/LiMn2O4 with a high voltage. J Electrochem Soc 158:A1490–A1497
Manickam M, Singh P, Issa T et al (2006) Cogeneration of cyclohexylamine and electrical power using PEM fuel cell reactor. J Appl Electrochem 36:599–602
Reiman KH, Brace KM, Gordon-Smith TJ et al (2006) Lithium insertion into TiO2 from aqueous solution. Electrochem Commun 8:517–522
Ding B, Xiao P, Ji G et al (2013) High-performance lithium-ion cathode LiMn0.7Fe0.3PO4/C and the mechanism of performance enhancements through Fe substitution. ACS Appl Mater Interfaces 5:12120–12126
Kim DK, Muralidharan P, Lee H-W et al (2008) Spinel LiMn2O4 nanorods as lithium ion battery cathodes. Nano Lett 8:3948–3952
Wan Z, Cai R, Jiang S et al (2012) Nitrogen and TiN-modified Li4Ti5O12: one-step synthesis and electrochemical performance optimization. J Mater Chem 22:17773–17781
Zhu Y, Wang C (2011) Novel CV for phase transformation electrodes. J Phys Chem C115:823–832
Sun D, Xue X, Tang YG et al (2015) High-rate LiTi2(PO4)3@N-C composite via Bi-nitrogen sources doping. ACS Appl Mater Interfaces 7:28337–28345
Acknowledgements
The authors gratefully acknowledge the staff in the Institute of Chemical Power Source of Soochow University for their assistance in various measurements. This study was funded by the National Natural Science Foundation of China (Grant No. 21203134 and 21671200) and the Prospective Project of Industry-University-Research Institution of Jiangsu Province, China (Grant No. BY2014059-13).
Funding
The support of this work from the National Natural Science Foundation of China (Grant No. 21203134 and 21671200) and the Prospective Project of Industry-University-Research Institution of Jiangsu Province, China (Grant No. BY2014059-13) is acknowledged.
Authors’ contributions
YY and HΒW conceived and designed the study. YY and HΒW performed the experiments. YY and DS wrote the paper. YY, DS, HBW, and HYW reviewed and edited the manuscript. All authors read and approved the manuscript.
Competing interests
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Yu, Y., Sun, D., Wang, H. et al. Electrochemical Properties of Rutile TiO2 Nanorod Array in Lithium Hydroxide Solution. Nanoscale Res Lett 11, 448 (2016). https://doi.org/10.1186/s11671-016-1662-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s11671-016-1662-8