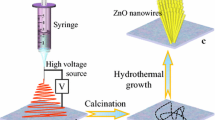
Abstract
Structural color was generated using electrospinning and hydrothermal growth of zinc oxide (ZnO). An aligned seed layer was prepared by electrospinning, and the hydrothermal growth time control was adjusted to generate various structural colors. The structural color changed according to the angle of the incident light. When the light was parallel to the direction of the aligned nanofibers, no pattern was observed. This pattern is referred to as an “optical switching pattern.” Replication using polydimethylsiloxane (PDMS) also enabled the generation of structural colors; this is an attractive approach for mass production. Additionally, the process is quite tunable because additional syntheses and etching can be performed after the patterns have been fabricated.
Similar content being viewed by others
Background
Structural color has many advantages over pigment (chemical) color. For example, it may be eco-friendly and does not suffer from photochemical degradation. Also, because the color changes according to the observing angle, it is possible to produce various patterns that cannot be produced with conventional pigment colors. These attributes have rendered structural colors of great interest to the textiles, paints, cosmetics, security, and sensors [1,2,3,4,5,6,7]. A variety of coloring principles explain the expression of structural color, and recent studies have shown that zinc oxide (ZnO) nanostructures express color by quasi-ordered scattering [8].
Quasi-ordered scattering is determined by the size and spacing of the nanostructures and is colored when the size of the nanostructure is similar and the spacing is constant. Although the diffuse reflectance is presumed to be the main coloring principle of quasi-ordered scattering, the principle of precise coloring has not yet been clarified, and blue, green, and purple are mainly observed [8].
A seed layer is required to fabricate ZnO nanostructures. Hydrothermal growth occurs in the region where the seed layer forms, which is also where structural color is expressed [9,10,11,12,13,14]. Hydrothermal growth refers to the synthesis of nanostructures in water at 40–80 °C. Therefore, the shape of the pattern is defined by the region of the seed layer. To fabricate optical switching patterns, a nanofiber seed layer is required that is aligned in one direction. To accomplish this, we used electrospinning, which is the most commonly used method for fabricating nanofibers [15,16,17,18]. However, collected electrospun nanofibers are usually randomly aligned. Research has been conducted to align nanofibers to minimize the net torque of electrostatic forces applied to the fiber ends [19]. In this way, the nanofibers can be aligned in a floating state (the nanofibers are aligned in the air between the electrodes), and an aligned seed layer can be fabricated by transferring the fabricated nanofibers to the target substrate. In order to produce the wire pattern of microscale without using electrospinning, a complicated patterning process using photoresist must be performed, which is a process that is not only difficult to realize mass production and large-scale as well as increase the process cost.
The fabricated seed layer was made from nanofibers having specific dimensions obtained through hydrothermal growth after heat treatment. ZnO is a highly suitable material for fabricating patterns because of its high refractive index (n = 2.0034) and ease of synthesis in various forms. The method of fabrication of structural color patterns using aligned ZnO nanofibers proposed in this study can be applied to create visual patterns, or in sensors for detecting various gases [20,21,22].
Experimental Methods
Materials
Polyvinylpyrrolidone (PVP; AR grade, M.W. 1,300,000) powder was purchased from Alfa Aesar. Ammonia solution (AR grade, 28.0–30.0% (mol/mol)), zinc chloride (AR grade), and zinc nitrate hexahydrate (AR grade) were purchased from Junsei Chemical Co., Ltd. Hydrochloric acid (AR grade) and N,N-dimethylformamide (DMF; AR grade) were purchased from Sigma–Aldrich. All reagents were used as-received and without further purification.
Electrospinning Conditions
Electrospinning was performed at room temperature and low humidity (relative humidity, 15–20%). A solution in DMF of 500 mM Zn(NO3)2 and 0.2 g/mL of PVP (final concentrations) was prepared. The gap between the tip and collector was fixed at 50 mm, and the applied voltage was 6.5 kV. To obtain aligned microwires, parallel aluminum electrodes were fabricated with dimensions of 3 cm in width and 2 cm in height. The nanofibers collected in parallel by an electric field were transferred to a target substrate (glass or silicon wafer).
ZnO Nanostructure Fabrication
To fabricate a ZnO nanostructure that exhibits structural color, a ZnO seed layer must be prepared by heat treatment (500 °C) of the nanofibers prepared in the previous step. Hydrothermal growth was then used to fabricate nanostructures on the seed layer. To fabricate the ZnO nanostructures, ZnCl2 was dissolved in deionized water (DI) at a concentration of 10 mM and maintained at 40–80 °C to initiate the reaction. Ammonia (NH4OH) was added to this aqueous solution at a rate of 5 μL/mL, generating OH− and raising the pH of the solution. In this environment, the Zn2+ ions quickly precipitated out of solution, which led to the nucleation and growth of ZnO nanostructures. To induce nanostructure synthesis at a constant rate, the reaction was carried out at pH > 10, and the pH of the solution decreased due to a dehydration reaction. Hydrothermal growth can be achieved by further growth of the nanostructures after patterning.
Patterning of ZnO Microwires
The growth of the nanostructures can be adjusted by using lithography to alter the time during which the seed layer is exposed to the reaction solution. In this study, lithography was performed with the help of masking tape. The masking tape was patterned using a paper cutter (Silhouette Cameo) to cut it into the desired shapes.
Characterization
The morphology of the ZnO nanostructures was observed by scanning electron microscopy (SEM) using a TESCAN LYRA 3 XMH instrument. Microwires were studied using an optical microscope (model D800; Nikon) equipped with a digital camera (model LV-150; Nikon). A white LED was used as the light source.
Replication of Pattern Using PDMS
The final fabricated ZnO nanostructure is used as a master mold for replication. Replication is carried out using polydimethylsiloxane (PDMS), which is characterized by being inexpensive, flexible, and optically transparent. First, pre-polymer base is mixed with curing agent 10: 1 and bubbles are removed in a vacuum chamber for 1 h to remove bubbles. Pour over the master mold and cure for 1 h at 65 °C in the oven to complete the replication process.
Results and Discussion
Aligned nanofibers are required to produce an optical switching pattern. Nanofibers floating in air are aligned using the parallel collector described above and then transferred to the target substrate (Fig. 1a). The aligned nanofibers on the target substrate are then heat-treated using hot plate (500 °C) to decompose the polymer component and form a thin ZnO seed layer (Fig. 1b). This layer can be grown hydrothermally to obtain the desired structural colors, and the part where the hydrothermal growth occurs can be controlled by patterning the reaction area using a masking technique (Fig. 1c). Then, final pattern is obtained by removing masking tape or additional patterning can be conducted by additional patterning and hydrothermal growth.
Schematic illustration of the aligned zinc oxide (ZnO) structural color fabrication process. a The electrospun nanofiber is collected in a vertical direction between parallel electrodes and transferred to the target substrate. b To remove the polymer component of the transferred nanofiber, heat treatment is performed at 500 °C to form a seed layer. c Patterning is performed using masking tape, and hydrothermal growth is performed in a constant temperature bath. d Removing the masking tape completes the final pattern. (Additional masking and hydrothermal growth allow complex patterns to be created)
Figure 2 shows the structural color obtained by varying the hydrothermal growth time of the microwires. As the hydrothermal growth time increases, the thickness of the microwire increases, which causes the optical properties to change. Figure 2a shows the hydrothermal growth time increasing from left to right by 2 min, and the bottom image shows a sample grown for four additional minutes. The structural colored pattern was reproducible for a given synthesis time, and the reaction region was localized using the masking method. Figure 2b shows a sample made to fabricate the sample with randomly bright structural colors. To generate the random colors, a sample with a seed layer was submerged randomly to the hydrothermal growth solution by shaking the sample or spraying the hydrothermal growth solution on the substrate. A random color sample resulted, free of a masking line. The lower SEM image demonstrates that microwires of various dimensions were produced with various colored segments.
Figure 3 shows how the techniques based on this ZnO microwire fabrication method can be extended. The process of making a structural color using ZnO microwires is not disadvantageous to mass production. The simplest way to mass-produce is to use molds. Figures 3A and A’ show patterns produced using ZnO nanostructured patterns on a glass substrate and duplicated patterns using polydimethylsiloxane (PDMS), respectively. In the replicated pattern using PDMS, the shape of ZnO nanostructure is replicated intact in PDMS (ZnO nanostructure remains on the original glass substrate and does not transferred to PDMS pattern). Figure 3A is a pattern made on glass, while Fig. 3A’ is one made with PDMS; both were fabricated on a transparent substrate. Also, Fig. 3A is an optical image of a sample that has undergone replication 10 times. This confirms that the pattern is well fabricated during the repetitive replication process. In that way, we could observe the structural color when the light coming from the back penetrated the pattern. Since light must pass through the pattern, the transparent substrate must be illuminated from the back, but the light source, the pattern, and the detector to observe do not have to be in a line. The structural color observed in the duplicated sample was similar. Figure 3B shows a sample that demonstrated structural color change through additional growth by restricting the portion to be grown after constructing the structural color. The colors are clearly different from each other. Figure 3B’ shows the result of close examination of the part labeled B′ in Fig. 3B with an optical microscope. Most of the nanofibers are well-aligned in the vertical direction. Clear boundaries are visible between the yellow-colored outer part of the circle indicated by C and the green-colored inner part of the circle indicated by D. Figure 3C, D shows SEM images of C and D, respectively. Further synthesis led to an increase in the overall microwire dimension, but the change in the size of each nanostructure constituting the microwire caused the change in structural color. The SEM image shows that the size of each nanostructure has also been increased, which causes the quasi-ordered scattering.
a Structural color pattern of an angel and the pattern duplicated 1 time (A’) and 10 times (A”) using polydimethylsiloxane. b Pattern for which two colors were obtained by varying the synthesis time and (b’) an image of the edge portion observed with an optical microscope. c, d Scanning electron microscope images of nanofibers in the outer and inner parts of b’
Structural color changes with viewing angle. Our structures displayed this feature. As noted above, the visible color of a transparent substrate differs from that of a reflecting substrate. With a transparent substrate, light is observed through the substrate, while with a reflecting substrate, light is reflected by the substrate and observed directly by our eyes. In both environments, the characteristic of changing color depending on the angle of observation was retained. Figure 4a shows structural color fabricated on a reflecting substrate (silicon wafer), and Fig. 4b shows structural color made on a transparent substrate (glass). It is evident that the structural color changed according to the angle of incidence. Moreover, not only did the color change with observing angle, but the alignment of the nanofibers enabled the pattern to be made brighter or invisible simply by changing the angle of incidence. If light is incident parallel to the alignment direction of the nanofibers, they hardly reflect the light. On the other hand, if light is incident perpendicularly, it is reflected in many directions, which makes the fiber array easy to see (Fig. 4c). Specifically, light incident in the perpendicular direction is incident on the entire cylindrical portion of the fiber surface, which results in clear visibility because it is reflected in a very wide direction. On the other hand, light incident in the parallel direction can only reflect in a limited direction, so that the total amount of light emitted is inevitably small, making it invisible.
Change of color of a structural pattern as a function of incidence angle on a a reflecting substrate and b transparent substrate. c Effect on pattern visibility by the orientation of incident light relative to the alignment direction of the nanofibers. Left: perpendicular, right: parallel orientation
Conclusion
We fabricated an optical switching pattern using ordered structural coloring nanostructures. The fabricated nanostructures are colored according to the principle of quasi-ordered scattering. Controlling the reaction time affects the size of the nanostructures and thereby the observable colors. We also used electrospinning, which is the most common method for fabricating nanofibers, to form an aligned seed layer to fabricate the alignment pattern. Our fabrication process is highly flexible, because the electrospinning process controlling the position and size of the pattern and the hydrothermal growth controlling the size of ZnO nanostructure can be modified independently. After the process is completed, the pattern can be modified by additional synthesis or etching, and the completed pattern can be mass-produced through replication using PDMS. Large color-changing patterned areas can be produced, for which the color changes according to the viewing direction and the light transmission direction. We successfully fabricated an optical switching pattern, for which the pattern was seen only on one side by aligning the nanofibers along one direction. We expect that our pattern-making method will find widespread applications in applications such as gas sensors and anti-tampering tags.
Abbreviations
- DI:
-
Deionized water
- PDMS:
-
Polydimethylsiloxane
- PVP:
-
Polyvinylpyrrolidone
- SEM:
-
Scanning electron microscopy
- ZnO:
-
Zinc oxide
References
Kim JH, Moon JH, Lee S-Y, Park J (2010) Biologically inspired humidity sensor based on three-dimensional photonic crystals. Appl Phys Lett 97:103701. https://doi.org/10.1063/1.3486115
Zhao Y, Zhao X, Tang B et al (2010) Quantum-dot-tagged bioresponsive hydrogel suspension array for multiplex label-free DNA detection. Adv Funct Mater 20:976–982. https://doi.org/10.1002/adfm.200901812
Sönnichsen C, Reinhard BM, Liphardt J, Alivisatos AP (2005) A molecular ruler based on plasmon coupling of single gold and silver nanoparticles. Nat Biotechnol 23:741–745. https://doi.org/10.1038/nbt1100
Parker AR, McPhedran RC, McKenzie DR et al (2001) Photonic engineering. Aphrodite’s iridescence. Nature 409:36–37. https://doi.org/10.1038/35051168
Kuo W-K, Weng H-P, Hsu J-J, Yu HH (2016) A bioinspired color-changing polystyrene microarray as a rapid qualitative sensor for methanol and ethanol. Mater Chem Phys 173:285–290. https://doi.org/10.1016/j.matchemphys.2016.02.014
Kuo W-K, Weng H-P, Hsu J-J, Yu HH (2016) Photonic crystal-based sensors for detecting alcohol concentration. Appl Sci 6:67. https://doi.org/10.3390/app6030067
Shieh J-Y, Kuo J-Y, Weng H-P, Yu HH (2013) Preparation and evaluation of the bioinspired PS/PDMS photochromic films by the self-assembly dip–drawing method. Langmuir 29:667–672. https://doi.org/10.1021/la303491c
Kim GH, An T, Lim G (2017) Bioinspired structural colors fabricated with ZnO quasi-ordered nanostructures. ACS Appl Mater Interfaces 9:19057–19062. https://doi.org/10.1021/acsami.6b15892
Al-lami S, Jaber H (2014) Controlling ZnO nanostructure morphology on seedless substrate by tuning process parameters and additives. Chem Mater Res 6:101–109
Baruah S, Dutta J (2009) Hydrothermal growth of ZnO nanostructures. Sci Technol Adv Mater 10:013001. https://doi.org/10.1088/1468-6996/10/1/013001
Long T, Yin S, Takabatake K et al (2008) Synthesis and characterization of ZnO nanorods and nanodisks from zinc chloride aqueous solution. Nanoscale Res Lett 4:247–253. https://doi.org/10.1007/s11671-008-9233-2
Na J-S, Gong B, Scarel G, Parsons GN (2009) Surface polarity shielding and hierarchical ZnO nano-architectures produced using sequential hydrothermal crystal synthesis and thin film atomic layer deposition. ACS Nano 3:3191–3199. https://doi.org/10.1021/nn900702e
Position and density control in hydrothermal growth of ZnO nanorod arrays through pre-formed micro/nanodots—IOPscience. http://iopscience.iop.org/article/10.1088/0957-4484/19/39/395602/meta. Accessed 5 Apr 2018
Density-controlled hydrothermal growth of well-aligned ZnO nanorod arrays—IOPscience. http://iopscience.iop.org/article/10.1088/0957-4484/18/3/035605/meta. Accessed 5 Apr 2018
Li D, Wang Y, Xia Y (2004) Electrospinning nanofibers as uniaxially aligned arrays and layer-by-layer stacked films. Adv Mater 16:361–366. https://doi.org/10.1002/adma.200306226
Li D, Babel A, Jenekhe SA, Xia Y (2004) Nanofibers of conjugated polymers prepared by electrospinning with a two-capillary spinneret. Adv Mater 16:2062–2066. https://doi.org/10.1002/adma.200400606
Zhang C-L, Lv K-P, Hu N-Y et al (2012) Macroscopic-scale alignment of ultralong Ag nanowires in polymer nanofiber mat and their hierarchical structures by magnetic-field-assisted electrospinning. Small 8:2936–2940. https://doi.org/10.1002/smll.201201353
Kim Bumjoo, Cho Seong J., An Taechang, et al (2012) Optical switching patterns using electrospun nanofiber array. Phys status solidi RRL – Rapid Res Lett 6:409–411. doi: https://doi.org/10.1002/pssr.201206326
Li D, Ouyang G, McCann JT, Xia Y (2005) Collecting electrospun nanofibers with patterned electrodes. Nano Lett 5:913–916. https://doi.org/10.1021/nl0504235
Ahmad R, Tripathy N, Jung D-U-J, Hahn Y-B (2014) Highly sensitive hydrazine chemical sensor based on ZnO nanorods field-effect transistor. Chem Commun 50:1890–1893. https://doi.org/10.1039/C3CC48197B
Ju S, Lee K, Janes DB et al (2005) Low operating voltage single ZnO nanowire field-effect transistors enabled by self-assembled organic gate nanodielectrics. Nano Lett 5:2281–2286. https://doi.org/10.1021/nl051658j
Chen M-J, Yang J-R, Shiojiri M (2012) ZnO-based ultra-violet light emitting diodes and nanostructures fabricated by atomic layer deposition. Semicond Sci Technol 27:074005. https://doi.org/10.1088/0268-1242/27/7/074005
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grants funded by the Korea government (MSIP) (NO. 2015R1A2A1A14027903; NO. 2018R1A2A2A05023037). The English in this document has been checked by at least two professional editors, both native speakers of English. For a certificate, please see: http://www.textcheck.com/certificate/cJpHky.
Availability of Data and Materials
All datasets are presented in the main paper.
Author information
Authors and Affiliations
Contributions
GHK carried out the experiment and prepared the manuscript. GHK, TA, and GL participated in the experiment and discussion of the results. TA and GL analyzed the data and helped modify the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kim, G.H., An, T. & Lim, G. Fabrication of Optical Switching Patterns with Structural Colored Microfibers. Nanoscale Res Lett 13, 204 (2018). https://doi.org/10.1186/s11671-018-2614-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s11671-018-2614-2